- Maximize data quality—TriMax and Topaz deactivations improve peak shapes and responses at trace levels.
- Achieve picogram-level sensitivity across a wide range of challenging GC-amenable pesticides.
- Reliably identify pesticides even in complex cannabis flower sample matrix.
Pesticide analysis is a continuous challenge for cannabis testing labs due to the changing regulatory landscape and the complexity of analytes and matrices. Regulations continue to fluctuate state-to-state, monitoring different analytes at different levels. In addition, pesticide chemistries vary considerably, and labs face a prevalence of harsh matrices ranging from plant material to edibles to concentrates and more. Diluting samples can be an effective strategy for extending instrument uptime because less matrix is introduced, but it means the target analytes are also present at lower levels, making them more challenging to detect.
For accurate, low-level analysis of pesticides in cannabis, a highly inert sample flow path is needed, so column quality becomes critical. If the column is not sufficiently inert, interactions between the analytes and imperfections (active sites) on the column surface can occur, which negatively affects peak shape and response. These problems are traditionally exacerbated at low levels, but they can be overcome by using an RMX-5Sil MS column. This column is an exceptionally inert, long-lasting, high-efficiency column that performs well for sensitive GC-MS/MS analyses. It is powered by the novel TriMax deactivation, which provides a robust interface between the polymer and capillary tubing. This results in a highly inert surface that resists degradation from thermal and matrix-related stressors, keeping the instrument running at maximum performance longer.
To demonstrate its performance, we subjected the RMX-5Sil MS column to low-level analysis of pesticides in cannabis flower—a notoriously difficult matrix. We paired the column with a Topaz inlet liner for maximum inertness throughout the sample flow path. To assess analytical results independent of sample prep efficiency (since techniques vary greatly lab to lab), cannabis flower extract (see chromatogram) was spiked post-extraction at 1.7 ng/mL with 30 pesticides covering a range of chemical functionalities. This level is comparable to 0.1 µg/g pesticide in cannabis flower and ultimately presented as 0.17 pg on-column.
Highly symmetrical peak shapes were obtained on the RMX-5Sil MS column, facilitating easy, accurate integration. Across all pesticides over triplicate injections, the average peak symmetry was 1.00 ± 0.14, showing that even at extremely low levels in a difficult matrix, the sample flow path is inert, and pesticide peaks are in the preferred 0.8-1.2 symmetry window. Sensitive systems, such as GC-MS/MS, require inert consumables to maintain maximum performance, and, as shown here, the RMX-5Sil MS column and Topaz liner are ideal for the analysis of pesticides in cannabis flower.
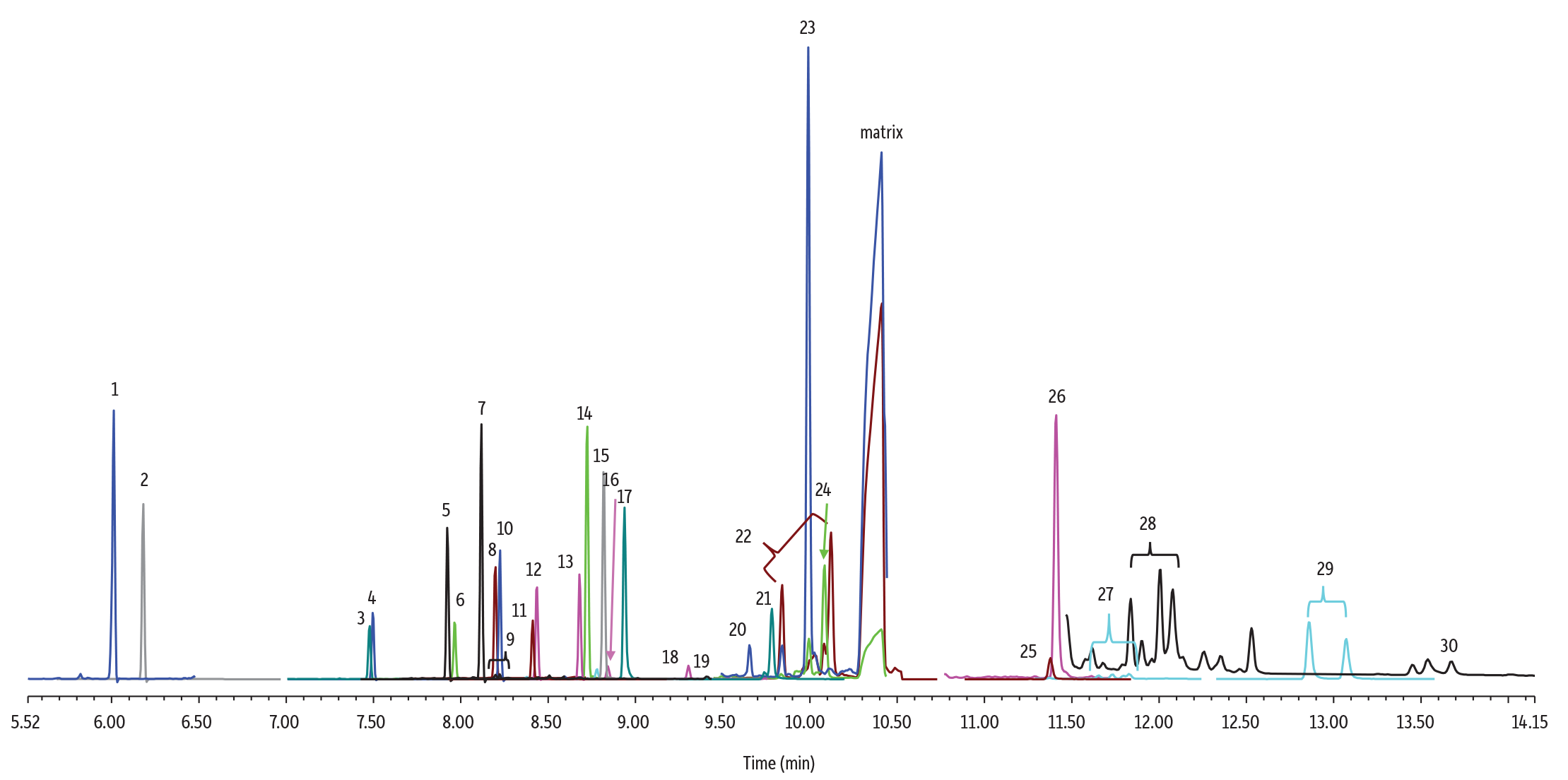
GC_FF1399
Peaks
| Peaks | tR (min) | Quant SRM | Qual SRM | |
|---|---|---|---|---|
| 1. | Mevinphos | 6.011 | 127.0 / 109.1 | 192.0 / 127.1 |
| 2. | Etridiazole | 6.178 | 211.0 / 183.0 | 182.9 / 140.0 |
| 3. | Pentachloronitrobenzene | 7.478 | 213.9 / 179.0 | 236.9 / 143.0 |
| 4. | Diazinone | 7.498 | 152.1 / 137.1 | 137.0 / 84.1 |
| 5. | Parathion methyl | 7.922 | 263.0 / 109.1 | 109.0 / 79.0 |
| 6. | Metalaxyl-m | 7.963 | 132.1 / 117.1 | 206.1 / 132.1 |
| 7. | Malathione | 8.118 | 127.1 / 99.0 | 173.1 / 117.1 |
| 8. | Chlorpyrifos | 8.194 | 197.0 / 169.0 | 314.0 / 257.9 |
| 9. | Allethrin | 8.219 | 123.1 / 122.1 | 79.1 / 77.1 |
| 10. | Fenthion | 8.224 | 278.0 / 109.0 | 278.0 / 245.1 |
| 11. | Fipronil | 8.409 | 213.0 / 143.0 | – / 212.9 |
| 12. | Cyprodinil | 8.434 | 224.1 / 222.2 | 210.1 / 208.1 |
| 13. | Tetrachlorvinphos | 8.68 | 328.9 / 109.1 | 109.0 / 79.0 |
| 14. | Paclobutrazol | 8.72 | 236.1 / 125.1 | 125.0 / 89.1 |
| 15. | Fludioxonil | 8.816 | 248.0 / 127.1 | 127.1 / 100.1 |
| Peaks | tR (min) | Quant SRM | Qual SRM | |
|---|---|---|---|---|
| 16. | Endosulfan I | 8.84 | 240.9 / 121.1 | 240.9 / 199.1 |
| 17. | Myclobutanil | 8.936 | 179.1 / 125.1 | 152.0 / 125.0 |
| 18. | Iprodione | 9.207 | 244.0 / 160.0 | 244.0 / 187.0 |
| 19. | Endosulfan II | 9.302 | 195.0 / 159.0 | 207.0 / 172.0 |
| 20. | Piperonyl butoxide | 9.729 | 176.1 / 91.1 | 176.1 / 146.1 |
| 21. | Tebuconazole | 9.739 | 250.1 / 125.1 | 250.1 / 153.1 |
| 22. | Tetramethrin | 9.941 | 164.1 / 107.1 | 123.1 / 81.1 |
| 23. | Bifenthrin | 9.991 | 181.1 / 165.1 | 166.1 / 165.1 |
| 24. | Phosmet | 10.072 | 160.0 / 77.1 | 133.0 / 77.1 |
| 25. | Pyridaben | 11.26 | 147.1 / 117.1 | 117.1 / 115.1 |
| 26. | Coumaphos | 11.369 | 226.0 / 163.1 | 109.0 / 81.0 |
| 27. | Cyfluthrin | 11.651 | 206.1 / 151.1 | 163.0 / 91.1 |
| 28. | Cypermethrin | 11.954 | 181.1 / 152.1 | 163.0 / 91.1 |
| 29. | Fenvalerate | 12.865 | 125.0 / 89.1 | 167.1 / 125.1 |
| 30. | Deltamethrin | 13.681 | 181.1 / 152.1 | 252.9 / 174.0 |
Conditions
| Column | RMX-5Sil MS, 30 m, 0.25 mm ID, 0.25 µm (cat.# 17323) |
|---|---|
| Standard/Sample | Canadian pesticide standards kit (cat.# 32592) |
| Diluent: | 1:1 hexane:acetone (1% acetic acid) |
| Conc.: | 1.7 ng/mL (0.17 pg on-column) |
| Injection | |
| Inj. Vol.: | 1 µL split (split ratio 10:1) |
| Liner: | Topaz 5.0 mm ID Precision inlet liner w/wool (cat.# 23327) |
| Inj. Temp.: | 280 °C |
| Oven | |
| Oven Temp.: | 50 °C (hold 1 min) to 270 °C at 30 °C/min to 300 °C at 5 °C/min (hold 0.5 min) |
| Carrier Gas | He, constant flow |
| Flow Rate: | 1.4 mL/min @ 50 °C |
| Detector | TSQ 9000 MS/MS |
|---|---|
| Transfer Line Temp.: | 300 °C |
| Aperture Size: | 6 mm ID |
| Source Temp.: | 300 °C |
| Tune Type: | PFTBA |
| Ionization Mode: | EI |
| Instrument | Thermo Trace GC |
| Sample Preparation | Cannabis flower extract was spiked at 1.7 ng/mL with select pesticides featured in the Canadian pesticides standards kit (cat.# 32592). Extracts represent a 1:5 ratio of cannabis flower in acetonitrile (1% acetic acid) further diluted in hexane and acetone. Concentration reflects approximately 0.1 µg/g pesticide in flower. |







