Key Highlights
- Robust, high-throughput methods challenged with disease state urine with heightened potential for matrix effects.
- Effective separation of EtG and EtS from urinary interferences in a fast analysis with no need for column preconditioning.
- Includes a method optimized for EtG/EtS alone and an alternative method for combined EtG/EtS and barbiturates analysis.
Abstract
Ethyl glucuronide (EtG) and ethyl sulfate (EtS) are widely recognized urinary biomarkers for monitoring alcohol consumption. While their analysis offers key advantages, such as a multiday detection window and clinical specificity, it also presents significant challenges due to their highly polar nature. These issues are further exacerbated by isobaric interferences present in urine and the inherent variability of human urine. In this study, we present a robust, high-throughput method for LC-MS/MS analysis of EtG/EtS in urine. The method achieves consistent retention, resolves matrix interferences, and eliminates the need for extensive column preconditioning. Developed using normal, disease state, and synthetic urine, this method demonstrates strong performance in challenging matrices and is suitable for routine use in forensic and clinical testing labs. An alternative method is also presented that allows combined analysis of EtG and EtS along with barbiturates for more efficient toxicology lab workflows.
Introduction
Ethyl glucuronide (EtG) and ethyl sulfate (EtS) are established biomarkers for alcohol consumption. The analysis of EtG and EtS in urine offers many advantages for abstinence monitoring, including a three-day detection window, good stability in properly stored specimens, and clinical specificity. However, the analysis of these compounds poses challenges due to their polar nature, which makes them difficult to retain by reversed-phase chromatography. In addition, the presence of isobaric matrix interferences in urine requires full resolution of the target analytes and matrix components or else matrix suppression or enhancement can occur, which may prevent labs from achieving accurate results at the required detection limits. Further complicating this analysis is the inherently variable nature of human urine chemistry, which can differ in pH, specific gravity, and salt concentration.
Current methods to overcome the challenges with LC-MS/MS analysis of EtG/EtS include the use of ion-exchange chromatography. This can be very effective when alcohol metabolites are the only target analytes, but with ion-exchange methods the column needs to be preconditioned with an extensive number of matrix injections to ensure proper performance. As an alternative, in this study an optimized, reversed-phase method that does not require time-consuming column preconditioning was developed for EtG and EtS analysis. In addition, the use of reversed-phase chromatography enabled the development of a combined method for EtG/EtS and barbiturates, which are also analyzed in negative-ion mode. This consolidated approach can benefit toxicology labs by reducing cycle times associated with polarity-switching methods that accommodate barbiturates along with analytes analyzed in positive-ion mode. By removing barbiturates monitored in negative ESI from positive-ion mode methods, the instrument can dedicate more time to adequately sampling peaks without the added complexity of polarity switching.
The two reversed-phase methods presented here, which are based on a Force Biphenyl column, offer the retention necessary to reliably separate target analytes from matrix interferences and do not require column preconditioning. Importantly, both methods were developed using normal, disease state, and synthetic urine because using various urine types better represents the wide array of samples encountered during routine analysis. Disease state samples can be particularly problematic due to their enhanced matrix effects, so it is imperative that they are tested during method development to ensure method robustness. The alternative methods established in this study are rugged enough to satisfy the demands of high-throughput labs conducting LC-MS/MS analysis of EtG/EtS alone and also offer labs the opportunity to improve productivity by combining EtG, EtS, and barbiturates analysis into a single method.
Experimental
Calibration Standards and Quality Control Samples
Synthetic urine (UTAK) was fortified with EtG and EtS to prepare the calibration standards. The linearity range was constructed using nine points from 30-2000 ng/mL. Quality control (QC) samples were prepared in single donor human urine and included two different lots obtained from chronic kidney disease patients. Three QC levels were prepared at LQC (100 ng/mL); MQC (400 ng/mL); and HQC (1600 ng/mL).
In order to remove problematic matrix components, protein precipitation was performed followed by dilution with water. Urine (50 µL) was aliquoted to a 2 mL centrifuge tube and mixed with isotopically labeled internal standards (10 µL of 2500 ppb EtS-d5 and 5000 ppb EtG-d5 prepared in 0.1% formic acid in water). Cold acetonitrile (150 µL) was added to the tube, vortexed for ~30 seconds and centrifuged for 10 minutes at 4200 rpm. The supernatant (100 µL) was aliquoted to an autosampler vial and water (900 µL) was added before the sample was capped, vortexed for ~30 seconds, and 10 µL injected onto the LC-MS/MS for analysis.
Instrument Conditions
LC-MS/MS analysis of EtG/EtS in urine and synthetic urine samples was performed on a Shimadzu 8060 LC-MS/MS. The instrument conditions are presented in Table I, and Table II shows the ions that were monitored for each compound.
Table I: LC-MS/MS Instrument Conditions
| Column | Force Biphenyl 100 mm, 3.0 mm ID, 3 µm (cat.# 962931E) | ||
| Guard Column | Force Biphenyl EXP guard cartridge 5 x 3.0 mm ID (cat.# 962950253); an UltraShield UHPLC precolumn filter 0.2 µm frit (cat.# 25811) was installed before the guard cartridge. | ||
| Column Temperature | 30 °C | ||
| Injection Volume | 10 µL | ||
| Mobile Phase A | Water, 0.1% formic acid | ||
| Mobile Phase B | Methanol, 0.1% formic acid | ||
| Detector | ESI (-) MS/MS | ||
| Valve Position | Time (min) | Flow Rate (mL/min) | %B |
| Waste | 0.00 | 0.8 | 0 |
| MS | 0.50 | 0.8 | – |
| MS | 1.74 | 0.8 | 100 |
| Waste | 1.75 | 1.0 | – |
| Waste | 3.50 | 1.0 | 100 |
| Waste | 3.51 | 0.8 | 0 |
| Waste | 5.00 | 0.8 | 0 |
Table II: Ion Transitions for LC-MS/MS Analysis of EtG/EtS
| Analyte | Retention Time (min) | Precursor Ion | Product Ion 1 | Product Ion 2 |
|---|---|---|---|---|
| EtS | 1.34 | 125.0 | 96.8 | 80.1 |
| EtS-d5 | 1.34 | 130.0 | 97.9 | |
| EtG | 1.54 | 221.1 | 75.1 | 85.2 |
| EtG-d5 | 1.54 | 226.0 | 75.0 |
Results and Discussion
Method Development
Column Choice
Column choice is a critical part of method development, and particle size, dimensions, and stationary phase should all be carefully considered relative to analytical goals. The use of a fully porous particle column, such as Force columns, gives greater surface area than its superficially porous counterpart, and thus it provides additional analyte retention and greater resolution of the analytes from urine matrix interferences. In addition, the Force Biphenyl column offers superior robustness over multimode columns, which can require extensive preconditioning.
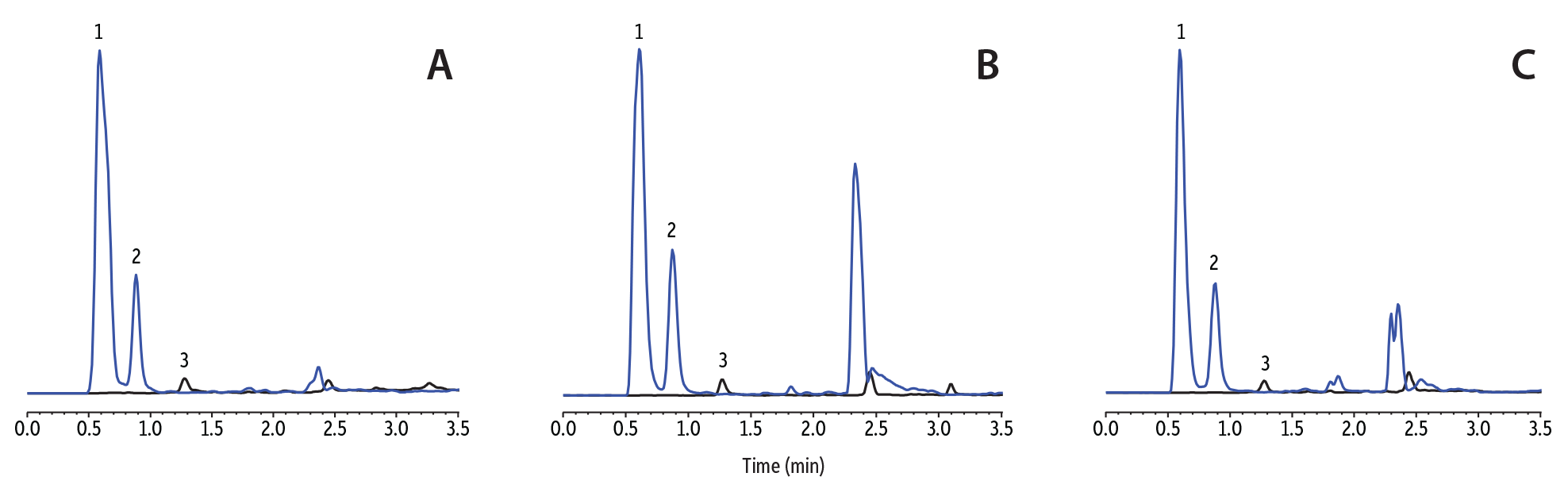
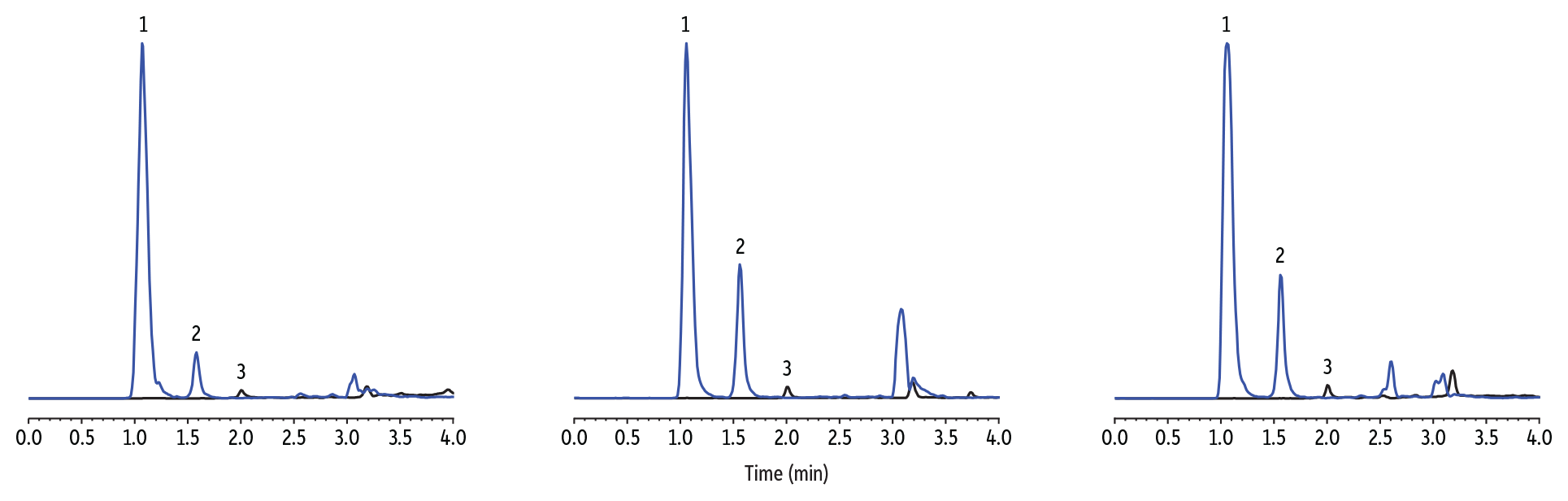
To select column dimensions, two Force Biphenyl column formats were assessed using a 50-ppb fortified chronic kidney disease (CKD) sample that was diluted 1:10. The method was developed using three different lots of CKD urine because it tends to have enhanced matrix effects compared to normal urine. As shown in Figure 1, the 50 x 3.0 mm, 3 µm column did not completely separate EtS from the urinary matrix interference. However, baseline resolution was achieved across all three CKD urine lots using the 100 x 3.0 mm, 3 µm Force Biphenyl column (Figure 2), so it was employed for subsequent experiments.
Ion Suppression/Enhancement Study
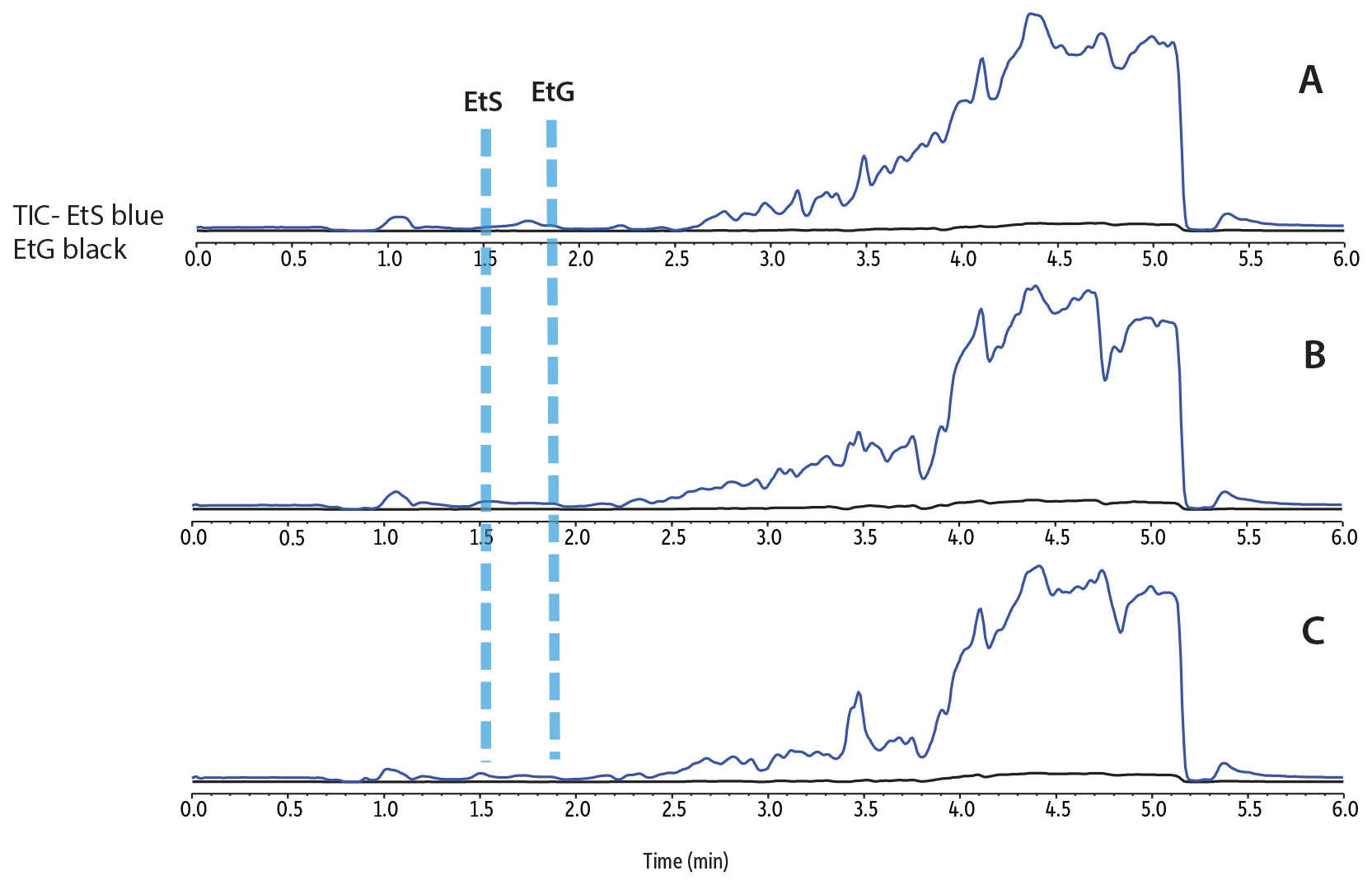
A study was performed to determine if the target analytes were eluting in regions where matrix components were also eluting but not appearing in the chromatogram because they do not share the same ion transitions as the target analytes. If the target analytes elute in these same regions, matrix components can compete with them for ionization and result in signal enhancement or suppression.
The potential for matrix effects was evaluated using post-column infusion of 1 ppm of EtG and EtS. While the infusion was being performed, a blank CKD urine sample diluted 1:10 was injected using the developed analytical method. In this test, spikes or dips in baseline correspond to matrix enhancements or suppression, respectively. As shown in Figure 3, the indicated elution times for both EtG and EtS show that both target analytes elute outside regions of significant matrix effects.
Carryover Assessment and Flow Settings
Several experiments were performed to ensure that all urinary contaminants were washed from the column and no carryover was observed. Flow rate changes and switching valves were used to keep the method as concise as possible, while still performing adequate flushing and re-equilibration. The switching valve was set to redirect the eluting solvent from the analytical column to waste for the first half minute of the run so any unretained salts present in the urine would not be directed to the MS. After the elution of both target analytes, the valve was switched back to waste to direct any contaminants eluting from the analytical column to waste, and the flow was increased to 1 mL/min. The flow rate was then returned to the analytical separation conditions, and the starting conditions were restored for equilibration for the next injection.
Chromatographic Performance
The final method for LC-MS/MS analysis of EtG/EtS in human urine is shown in Figure 4 for a representative sample of CKD human urine spiked at the LQC level (100 ng/mL). Good retention and peak shape were observed for both analytes on the Force Biphenyl column, and EtS was fully resolved from a potentially problematic matrix interference. These conditions were used in subsequent accuracy and precision experiments and as the basis of a consolidated method that also included barbiturates.
LC_CF0843
Peaks
| Peaks | tR (min) | Conc. (ng/mL) | Precursor | Product 1 | Product 2 | |
|---|---|---|---|---|---|---|
| 1. | Urinary interference | 0.89 | – | 125.0 | 80.1 | – |
| 2. | EtS-d5 | 1.34 | 1000 | 130.0 | 97.9 | – |
| 3. | EtS | 1.34 | 100 | 125.0 | 96.8 | 80.1 |
| 4. | EtG-d5 | 1.54 | 2000 | 226.0 | 75.0 | – |
| 5. | EtG | 1.54 | 100 | 221.1 | 75.1 | 85.2 |
Conditions
| Column | Force Biphenyl (cat.# 962931E) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 100 mm x 3 mm ID | ||||||||||||||||||
| Particle Size: | 3 µm | ||||||||||||||||||
| Pore Size: | 100 Å | ||||||||||||||||||
| Guard Column: | Force Biphenyl EXP guard cartridge 5 mm, 3 mm ID, 3 µm (cat.# 962950253) | ||||||||||||||||||
| Temp.: | 30 °C | ||||||||||||||||||
| Standard/Sample | |||||||||||||||||||
| Ethyl sulfate sodium salt (EtS) standard (cat.# 34103) | |||||||||||||||||||
| Ethyl sulfate-d5 sodium salt (EtS-d5) standard, 1000 µg/mL, methanol, 1 mL/ampul (cat.# 34104) | |||||||||||||||||||
| Ethyl-beta-D-glucuronide (EtG) standard, 1000 µg/mL, methanol, 1 mL/ampul (cat.# 34101) | |||||||||||||||||||
| Ethyl-beta-D-glucuronide-d5 (EtG-d5) standard, 1000 µg/mL, methanol, 1 mL/ampul (cat.# 34102) | |||||||||||||||||||
| Diluent: | Water | ||||||||||||||||||
| Conc.: | 100 ng/mL in urine | ||||||||||||||||||
| Inj. Vol.: | 10 µL | ||||||||||||||||||
| Mobile Phase | |||||||||||||||||||
| A: | Water, 0.1% formic acid | ||||||||||||||||||
| B: | Methanol, 0.1% formic acid | ||||||||||||||||||
| |||||||||||||||||||
| Flow: | *0.8 mL/min | ||||||||||||||||||
| Max Pressure: | 427 bar |
| Detector | Shimadzu 8060 MS/MS |
|---|---|
| Ion Source: | Electrospray |
| Ion Mode: | ESI- |
| Mode: | MRM |
| Instrument | Shimadzu Nexera X2 |
| Sample Preparation | Urine (50 μL) was aliquoted to a 2 mL centrifuge tube, fortified at 100 ng/mL with EtG and EtS, and mixed with isotopically labeled internal standards (10 μL). Cold acetonitrile (150 μL) was added to the tube, vortexed for ~30 seconds, and centrifuged for 10 minutes at 4200 rpm. The supernatant (100 μL) was aliquoted to an autosampler vial (cat.#21143), and water (900 μL) was added before the sample was capped (cat.#24498), vortexed for ~30 seconds, and 10 μL injected onto the LC-MS/MS for analysis. |
| Notes | An Ultra Shield UHPLC PreColumn Filter 0.2 μm frit (cat.# 25811) was installed before the guard cartridge. Valve position was directed to waste from 0.00-0.50 minutes and after 1.75 minutes. *Flow rate was adjusted to 1 mL/min from 1.75 minutes to 3.50 minutes to flush contaminates from the analytical column to reduce matrix effects. Flow rate was switched back to 0.8 mL/min at 3.51 min. |
Linearity, Accuracy, and Precision
Calibration studies showed good linearity across the entire range with r2 values of 0.995 or greater using 1/x weighted quadratic regression. QC samples were prepared over three days and analyzed in triplicate (n=9) for method accuracy and precision experiments, and Table III summarizes the results. Intraday percent recoveries range from 85.1-114.6 % with %RSD values of 9.6% or less. Interday percent recoveries range from 91.9-111.9% with %RSD values of 10.1% or less.
Table III: Method Accuracy (% Recovery) and Precision (%RSD) for LC-MS/MS Analysis of EtG/EtS in QC Urine Samples
| Day 1 | |||||||
|---|---|---|---|---|---|---|---|
| LQC (100 ng/mL) | MQC (400 ng/mL) | HQC (1600 ng/mL) | |||||
| Analyte | Urine Sample# | % Recovery | %RSD | % Recovery | %RSD | % Recovery | %RSD |
| EtS | CKD 1 | 113.1 | 0.7 | 105.5 | 1.2 | 102.2 | 2.7 |
| CKD 2 | 113.7 | 4.2 | 105.8 | 2.4 | 98.4 | 2.0 | |
| 3 | 109.6 | 5.7 | 114.6 | 5.0 | 113.1 | 3.8 | |
| 4 | 96.4 | 2.6 | 100.9 | 7.2 | 90.1 | 0.9 | |
| 5 | 106.8 | 1.5 | 112.6 | 8.9 | 110.7 | 1.0 | |
| 6 | 113.5 | 3.5 | 107.7 | 2.2 | 107.1 | 1.8 | |
| synthetic | 100.2 | 2.4 | 94.0 | 3.2 | 92.1 | 0.6 | |
| EtG | CKD 1 | 103.9 | 2.5 | 102.8 | 6.7 | 97.1 | 6.0 |
| CKD 2 | 89.6 | 3.5 | 93.2 | 1.4 | 98.5 | 4.6 | |
| 3 | 109.3 | 3.2 | 107.4 | 4.1 | 102.3 | 5.4 | |
| 4 | 88.3 | 3.1 | 87.6 | 5.4 | 100.0 | 5.1 | |
| 5 | 91.4 | 1.5 | 96.2 | 9.5 | 97.6 | 4.2 | |
| 6 | 108.1 | 6.5 | 94.9 | 3.9 | 97.4 | 3.8 | |
| synthetic | 106.9 | 8.2 | 95.3 | 1.4 | 99.4 | 6.1 | |
| Day 2 | |||||||
| Analyte | Urine Sample# | LQC (100 ng/mL) | MQC (400 ng/mL) | HQC (1600 ng/mL) | |||
| % Recovery | %RSD | % Recovery | %RSD | % Recovery | %RSD | ||
| EtS | CKD 1 | 112.6 | 2.1 | 92.6 | 4.5 | 108.2 | 4.4 |
| CKD 2 | 112.7 | 5.8 | 102.3 | 9.0 | 105.1 | 1.5 | |
| 3 | 112.3 | 3.8 | 106.8 | 4.9 | 101.8 | 7.9 | |
| 4 | 102.5 | 0.3 | 90.9 | 5.1 | 105.5 | 4.7 | |
| 5 | 102.0 | 2.9 | 98.6 | 3.4 | 100.9 | 4.0 | |
| 6 | 112.8 | 2.3 | 110.1 | 3.1 | 102.5 | 3.9 | |
| synthetic | 100.5 | 2.2 | 90.9 | 1.2 | 101.1 | 3.7 | |
| EtG | CKD 1 | 108.7 | 9.9 | 93.5 | 4.2 | 90.7 | 3.8 |
| CKD 2 | 102.4 | 2.6 | 94.8 | 6.1 | 99.0 | 3.5 | |
| 3 | 113.6 | 6.0 | 94.0 | 7.0 | 110.7 | 6.6 | |
| 4 | 99.6 | 1.2 | 101.3 | 5.6 | 96.9 | 4.3 | |
| 5 | 94.2 | 6.7 | 93.2 | 3.1 | 94.0 | 9.6 | |
| 6 | 110.9 | 1.7 | 90.3 | 3.2 | 96.3 | 6.9 | |
| synthetic | 98.5 | 6.1 | 103.0 | 4.1 | 109.5 | 4.8 | |
| Day 3 | |||||||
| Analyte | Urine Sample# | LQC (100 ng/mL) | MQC (400 ng/mL) | HQC (1600 ng/mL) | |||
| % Recovery | %RSD | % Recovery | %RSD | % Recovery | %RSD | ||
| EtS | CKD 1 | 109.9 | 3.8 | 102.3 | 5.7 | 110.5 | 1.7 |
| CKD 2 | 108.9 | 4.4 | 98.1 | 2.2 | 109.7 | 1.0 | |
| 3 | 107.4 | 2.0 | 97.2 | 0.9 | 108.0 | 1.9 | |
| 4 | 105.0 | 5.1 | 95.8 | 2.4 | 107.5 | 1.9 | |
| 5 | 106.6 | 6.0 | 99.6 | 7.3 | 108.8 | 3.9 | |
| 6 | 105.8 | 5.6 | 101.3 | 7.2 | 110.5 | 3.0 | |
| synthetic | 94.3 | 4.5 | 90.7 | 7.8 | 92.8 | 1.8 | |
| EtG | CKD 1 | 101.3 | 8.6 | 85.1 | 5.0 | 95.5 | 1.9 |
| CKD 2 | 97.4 | 8.9 | 94.3 | 3.3 | 103.1 | 7.3 | |
| 3 | 99.3 | 3.6 | 98.9 | 5.5 | 99.3 | 3.6 | |
| 4 | 90.9 | 7.5 | 91.7 | 4.3 | 103.3 | 2.4 | |
| 5 | 94.3 | 2.6 | 102.8 | 3.2 | 94.3 | 2.6 | |
| 6 | 91.6 | 4.3 | 104.5 | 3.3 | 91.6 | 4.3 | |
| synthetic | 104.0 | 1.6 | 102.3 | 3.8 | 91.0 | 3.0 | |
| Interday | |||||||
| LQC (100 ng/mL) | MQC (400 ng/mL) | HQC (1600 ng/mL) | |||||
| Analyte | Urine Sample# | % Recovery | %RSD | % Recovery | %RSD | % Recovery | %RSD |
| EtS | CKD 1 | 111.9 | 1.5 | 100.1 | 6.7 | 106.9 | 4.0 |
| CKD 2 | 111.8 | 2.2 | 102.0 | 3.8 | 104.4 | 5.4 | |
| 3 | 109.8 | 2.3 | 106.2 | 8.2 | 107.6 | 5.3 | |
| 4 | 101.3 | 4.4 | 95.9 | 5.2 | 101.0 | 9.4 | |
| 5 | 105.1 | 2.6 | 103.6 | 7.5 | 106.8 | 4.9 | |
| 6 | 110.7 | 3.8 | 106.4 | 4.3 | 106.7 | 3.7 | |
| synthetic | 98.3 | 3.6 | 91.9 | 2.0 | 95.3 | 5.2 | |
| EtG | CKD 1 | 104.6 | 3.6 | 93.8 | 9.5 | 94.4 | 3.6 |
| CKD 2 | 96.5 | 6.7 | 94.1 | 0.9 | 100.2 | 2.5 | |
| 3 | 107.4 | 6.8 | 100.1 | 6.8 | 104.1 | 5.7 | |
| 4 | 92.9 | 6.4 | 93.5 | 7.5 | 100.1 | 3.2 | |
| 5 | 93.3 | 1.8 | 97.4 | 5.1 | 95.3 | 2.1 | |
| 6 | 103.5 | 10.1 | 96.6 | 7.5 | 95.1 | 3.2 | |
| synthetic | 103.1 | 4.1 | 100.2 | 4.2 | 100.0 | 9.3 | |
In addition to assessing linearity, accuracy, and precision, method performance was verified using a third-party reference material. EtG Plus Level 1 Quality Control and EtG Plus Level 2 Quality Control reference materials were provided by UTAK and prepared and analyzed by the outlined method. Each level was prepared in six replicates and analyzed over three days (n=18). The results of these experiments are given in Table IV.
Table IV: Accuracy and Precision Data for UTAK EtG Plus Level 1 and Level 2 Quality Control Reference Material
| UTAK EtG Plus Level 1 Urine Control | UTAK EtG Plus Level 2 Urine Control | ||||
|---|---|---|---|---|---|
| Analyte | EtG | EtS | EtG | EtS | |
| Verified Value (ng/mL) | 510 | 240 | 1800 | 800 | |
| Day 1 (n=6) | % Recovery | 98.3 | 97.5 | 106.2 | 106.6 |
| %RSD | 7.84 | 3.50 | 5.68 | 4.80 | |
| Calculated Concentration (ng/mL) | 502 | 234 | 1912 | 853 | |
| Day 2 (n=6) | % Recovery | 103.0 | 101.7 | 104.0 | 105.7 |
| %RSD | 8.63 | 7.29 | 4.85 | 2.82 | |
| Calculated Concentration (ng/mL) | 523 | 245 | 1823 | 831 | |
| Day 3 (n=6) | % Recovery | 103.3 | 100.0 | 101.9 | 101.2 |
| %RSD | 10.91 | 8.50 | 4.57 | 3.62 | |
| Calculated Concentration (ng/mL) | 524 | 240 | 1784 | 795 | |
| Interday (n=18) | % Recovery | 101.6 | 100.0 | 104.1 | 104.5 |
| %RSD | 2.75 | 2.13 | 2.07 | 2.78 | |
| Calculated Concentration (ng/mL) | 516 | 240 | 1840 | 826 | |
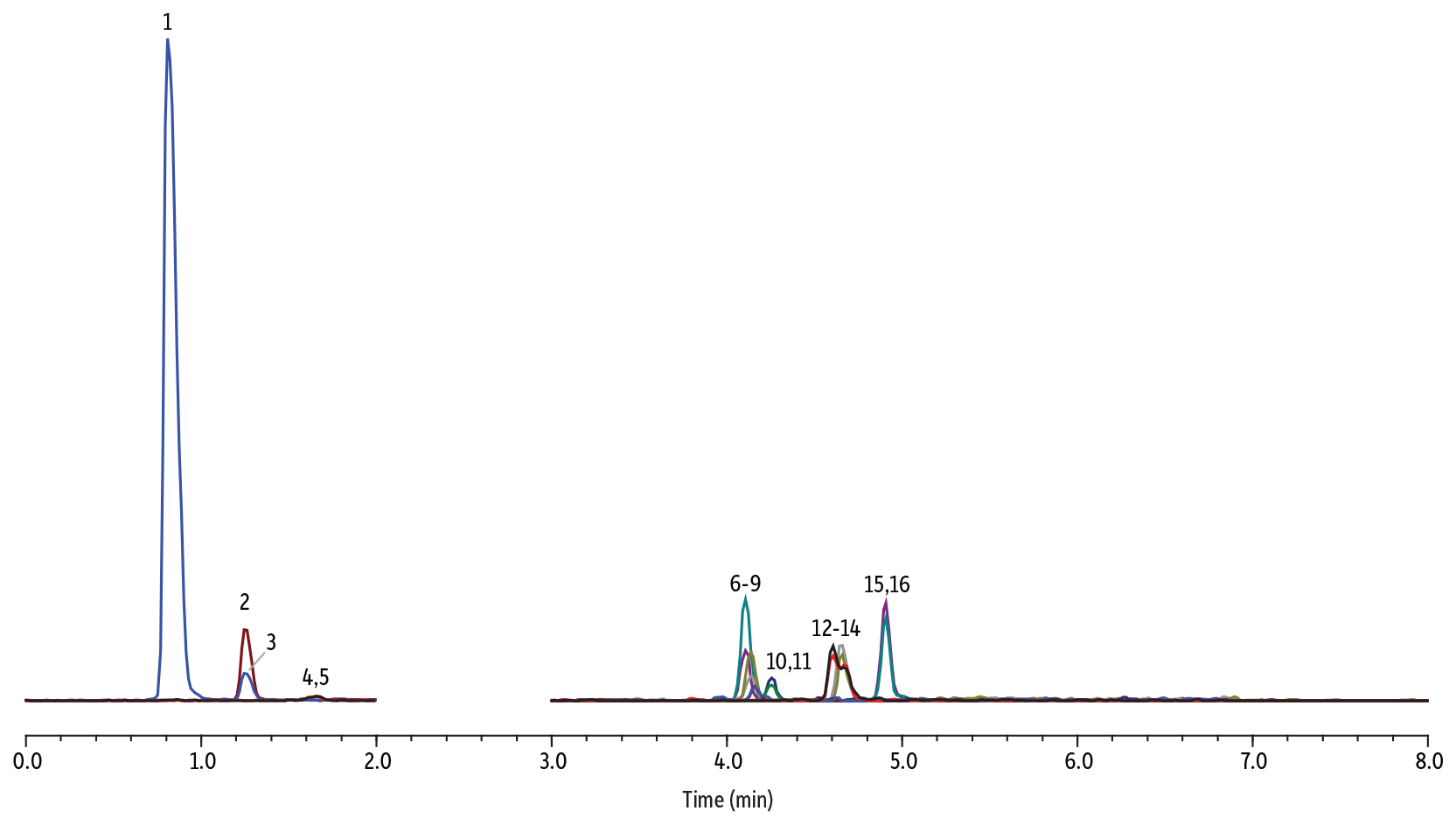
Development of a Consolidated Method for EtG, EtS, and Barbiturates
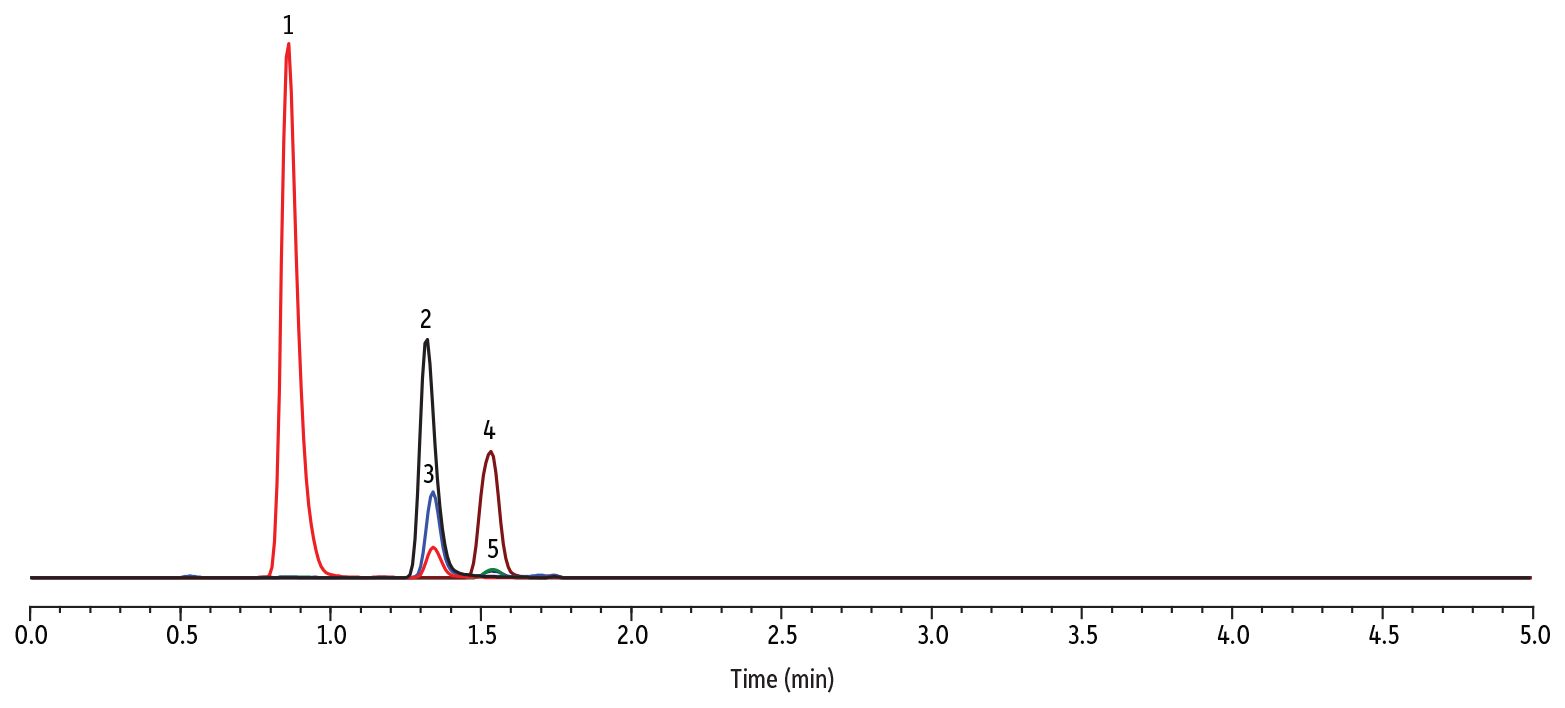
Barbiturates are another class of negative-mode compounds that are often analyzed in a separate panel. To allow labs to reduce column changes and utilize instrumentation more efficiently, a consolidated negative-mode panel was developed to allow quantitative analysis of both barbiturates and EtG and EtS alcohol metabolites. Instrument conditions and a representative chromatogram are given in Figure 5. It should be noted that amobarbital and pentobarbital are isomers but were not fully resolved for this method. Since they are only partially resolved, they are reported together here, but a secondary method, such as the one outlined in Figure 6, could be used to identify which isomer is present when needed.
LC_CF0847
Peaks
| Peaks | tR (min) | Conc. (ng/mL) | Precursor | Product 1 | Product 2 | |
|---|---|---|---|---|---|---|
| 1. | Urinary interference | 0.89 | – | 125.0 | 80.1 | – |
| 2. | EtS | 1.22 | 100 | 125.0 | 96.8 | 80.1 |
| 3. | EtS-d5 | 1.21 | 1000 | 130.0 | 97.9 | – |
| 4. | EtG | 1.65 | 100 | 221.1 | 75.1 | 85.2 |
| 5. | EtG-d5 | 1.64 | 2000 | 226.0 | 75.0 | – |
| 6. | Phenobarbital-d5 | 4.15 | 4000 | 236.0 | 42.0 | – |
| 7. | Phenobarbital | 4.18 | 200 | 231.2 | 42.1 | 188.1 |
| 8. | Butabarbital-d5 | 4.18 | 4000 | 216 | 41.9 | – |
| Peaks | tR (min) | Conc. (ng/mL) | Precursor | Product 1 | Product 2 | |
|---|---|---|---|---|---|---|
| 9. | Butabarbital | 4.19 | 200 | 211.0 | 168.3 | 42.0 |
| 10. | Butalbital-d5 | 4.23 | 4000 | 228.0 | 185.3 | – |
| 11. | Butalbital | 4.27 | 200 | 223.0 | 42.1 | 180.3 |
| 12. | Amobarbital | 4.63 | 200 | 225.0 | 182.3 | 42.1 |
| 13. | Amobarbital-d5 | 4.69 | 4000 | 230.0 | 42.0 | – |
| 14. | Pentobarbital | 4.71 | 200 | 225.0 | 182.3 | 42.1 |
| 15. | Secobarbital-d5 | 4.92 | 4000 | 242.0 | 199.3 | – |
| 16. | Secobarbital | 4.94 | 200 | 237.0 | 42.1 | 194.2 |
Conditions
| Column | Force Biphenyl (cat.# 962931E) | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 100 mm x 3 mm ID | ||||||||||||||||||||||||||||
| Particle Size: | 3 µm | ||||||||||||||||||||||||||||
| Pore Size: | 100 Å | ||||||||||||||||||||||||||||
| Guard Column: | Force Biphenyl EXP guard cartridge 5 mm, 3 mm ID, 3 µm (cat.# 962950253) | ||||||||||||||||||||||||||||
| Temp.: | 30 °C | ||||||||||||||||||||||||||||
| Standard/Sample | |||||||||||||||||||||||||||||
| Ethyl sulfate sodium salt (EtS) standard (cat.# 34103) | |||||||||||||||||||||||||||||
| Ethyl sulfate-d5 sodium salt (EtS-d5) standard, 1000 µg/mL, methanol, 1 mL/ampul (cat.# 34104) | |||||||||||||||||||||||||||||
| Ethyl-beta-D-glucuronide (EtG) standard, 1000 µg/mL, methanol, 1 mL/ampul (cat.# 34101) | |||||||||||||||||||||||||||||
| Ethyl-beta-D-glucuronide-d5 (EtG-d5) standard, 1000 µg/mL, methanol, 1 mL/ampul (cat.# 34102) | |||||||||||||||||||||||||||||
| Butalbital standard, 1000 µg/mL, P&T methanol, 1 mL/ampul (cat.# 34032) | |||||||||||||||||||||||||||||
| Phenobarbital standard, 1000 µg/mL, P&T methanol, 1 mL/ampul (cat.# 34037) | |||||||||||||||||||||||||||||
| Pentobarbital standard, 1000 µg/mL, P&T methanol, 1 mL/ampul (cat.# 34036) | |||||||||||||||||||||||||||||
| Secobarbital standard, 1000 µg/mL, P&T methanol, 1 mL/ampul (cat.# 34038) | |||||||||||||||||||||||||||||
| Diluent: | Water | ||||||||||||||||||||||||||||
| Inj. Vol.: | 10 µL | ||||||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||||||
| A: | Water, 0.1% formic acid | ||||||||||||||||||||||||||||
| B: | Methanol, 0.1% formic acid | ||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Max Pressure: | 427 bar |
| Detector | Shimadzu 8060 MS/MS |
|---|---|
| Ion Source: | Electrospray |
| Ion Mode: | ESI- |
| Mode: | MRM |
| Instrument | Shimadzu Nexera X2 |
| Sample Preparation | Urine (50 μL) was aliquoted to a 2 mL centrifuge tube, fortified at either 100 ng/mL for EtG and EtS or 200 ng/mL for barbiturates, and was mixed with isotopically labeled internal standards (10 μL). Cold acetonitrile (150 μL) was added to the tube, vortexed for ~30 seconds, and centrifuged for 10 minutes at 4200 rpm. The supernatant (100 μL) was aliquoted to an autosampler vial (cat.#21143) and water (900 μL) was added before the sample was capped (cat.#24498), vortexed for ~30 seconds, and 10 μL injected onto the LC-MS/MS for analysis. |
| Notes | An Ultra Shield UHPLC PreColumn Filter 0.2 μm frit (cat.#25811) was installed before the guard cartridge. |
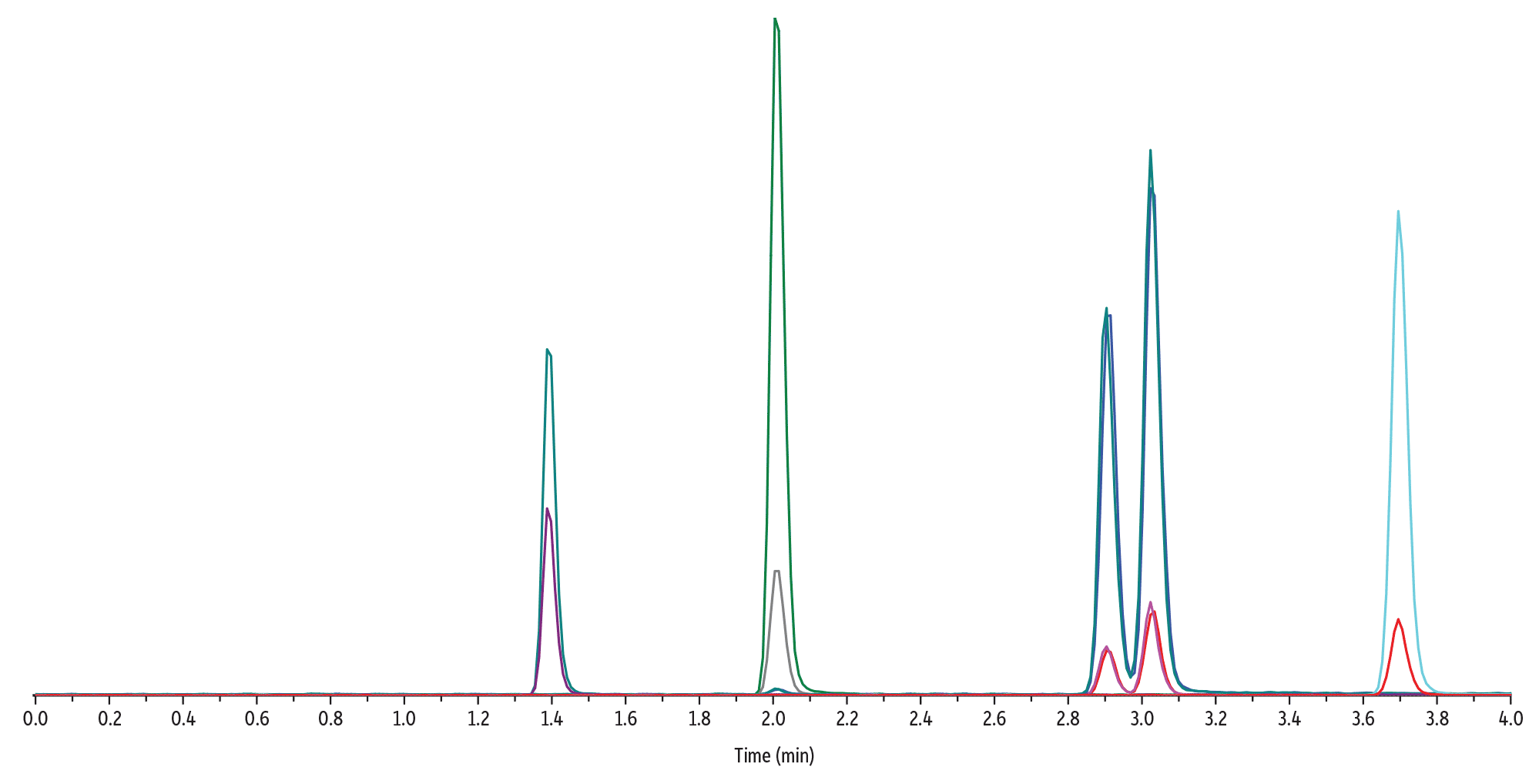
LC_CF0619
Peaks
| Peaks | tR (min) | Precursor Ion | Product Ion | Product Ion | |
|---|---|---|---|---|---|
| 1. | Phenobarbital | 1.39 | 230.9 | 187.8 | 85.0 |
| 2. | Butalbital | 2.01 | 223.0 | 180.0 | 84.9 |
| 3. | Pentobarbital | 2.91 | 225.0 | 182.0 | 84.9 |
| 4. | Amobarbital | 3.03 | 225.0 | 182.0 | 84.8 |
| 5. | Secobarbital | 3.71 | 237.0 | 193.9 | 84.8 |
Conditions
| Column | Raptor C18 (cat.# 9304A12) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 100 mm x 2.1 mm ID | ||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||
| Temp.: | 50 °C | ||||||||||||||||||||
| Standard/Sample | |||||||||||||||||||||
| Diluent: | Water | ||||||||||||||||||||
| Conc.: | 500 ng/mL | ||||||||||||||||||||
| Inj. Vol.: | 5 µL | ||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||
| A: | Water + 0.1% formic acid | ||||||||||||||||||||
| B: | Acetonitrile + 0.1% formic acid | ||||||||||||||||||||
|
| Detector | MS/MS |
|---|---|
| Ion Mode: | ESI- |
| Mode: | MRM |
| Instrument | UHPLC |
To evaluate performance of the consolidated method, linearity, accuracy, and precision were assessed. Calibration standards were prepared in synthetic urine (provided by UTAK) fortified with EtG and EtS alcohol metabolites and barbiturates. The linearity range was constructed using nine points from 30-2000 ng/mL for alcohol metabolites and 60-4000 ng/mL for barbiturates. Calibration results showed good linearity with r2 values of 0.992 or greater using 1/x weighted quadratic regression.
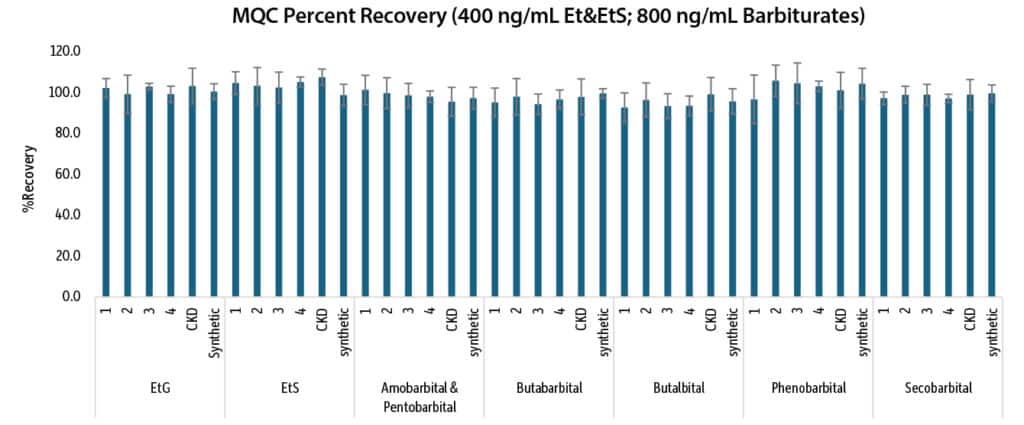
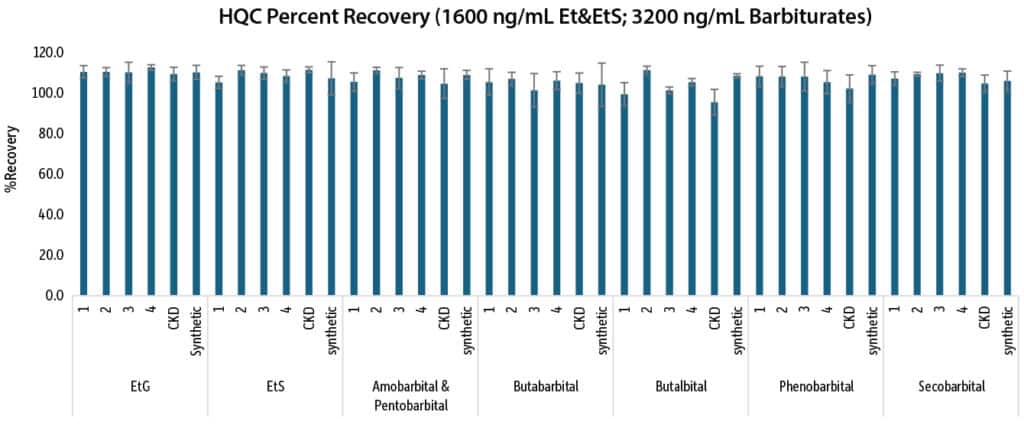
To assess accuracy and precision, QC samples were prepared over three days in triplicate in single donor human urine and included one lot that was obtained from a CKD patient. Three quality control levels were prepared at LQC (100 ng/mL); MQC (400 ng/mL); and HQC (1600 ng/mL) for the alcohol metabolites and at LQC (200 ng/mL); MQC (800 ng/mL); and HQC (3200 ng/mL) for the barbiturates.
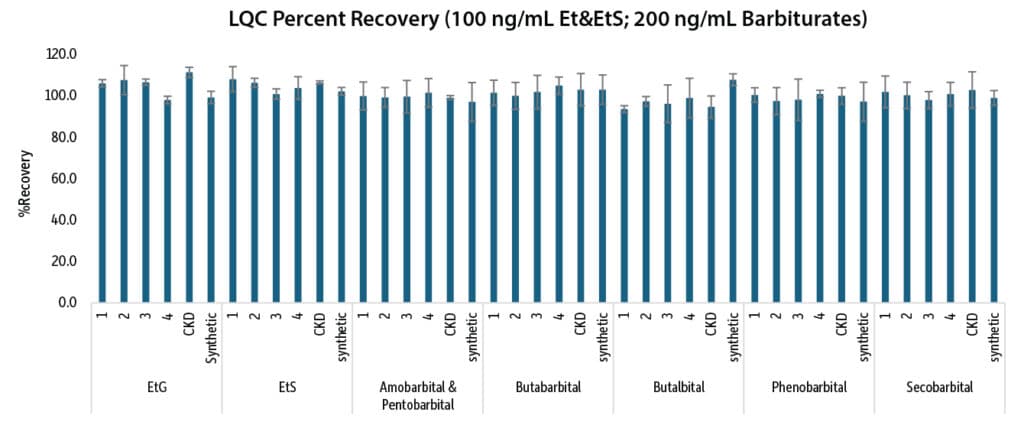
Figures 7, 8, and 9 summarize the interday accuracy and precision data for the consolidated method. The percent recovery range was 87.3-114.8% and %RSD were less than 10.7% for the intraday results. The interday percent recovery range was 92.7-112.9% with %RSD values of 11.9% or less.
Conclusion
The reversed-phase LC-MS/MS methods for analysis of EtG and EtS developed on the Force Biphenyl column successfully resolved matrix interferences from the analytes of interest across all evaluated urine lots, including challenging CKD samples. Two different methods were developed: a dedicated method that targets the alcohol metabolites alone, and a consolidated method for both alcohol metabolites and barbiturates, which can help labs optimize use of their instrumentation. Linearity was demonstrated for both methods with r² ≥ 0.99, and both methods exhibited acceptable intra- and interday precision and accuracy.
The methods presented here on the Force Biphenyl column do not require column preconditioning, and they were developed using a wide variety of urine types, including CKD urine, to ensure reliable performance across the wide variety of urine sample types commonly encountered during routine analysis. Use of the Force Biphenyl column for these methods provides labs with additional flexibility and efficiency because the column performs well for several related analysis, including drugs of abuse and novel psychoactive substances, which reduces the need to change columns when switching an instrument to a different method.
Acknowledgments
The authors wish to thank UTAK for providing synthetic urine as well as the EtG Plus Level 1 Quality Control and EtG Plus 2 Quality Control samples.
This method has been developed for research use only; it is not suitable for use in diagnostic procedures without further evaluation.