Move over, nitazenes! There’s a new NPS in town. In July of 2025, the Chinese government announced that nitazene analogs would be placed under national control, ultimately leading to a decline in positivity for these compounds. [1,2] It hasn’t taken long for a new subclass of designer drugs to take their place. Over the last several months, a group of synthetic opioids known as “orphine analogs” have been increasingly detected in toxicology and seized drug cases.
Despite the recent attention orphines have been garnering, this subclass of drugs has been around for some time. Some of these compounds, such as spirochlorphine, were originally synthesized for potential pharmaceutical use back in the 1970s. [3] The compound brorphine made headlines back 2020 after it was detected in several overdose cases but fell out of popularity after the U.S. DEA deemed it a controlled substance. [4] Much like the nitazene analogs, the orphines have started to evolve and new substances are emerging on the illicit drug market. While information on the pharmacology of these compounds is still limited, there is data that suggests that some of these compounds may be more potent than fentanyl. [2]
If your lab is considering adding one or more of these orphine analogs to your LC-MS/MS testing method, you’ve come to the right place. Below you will find the precursor and product ions for two frequently encountered orphines as well as where you might expect them to elute in a multi-drug class assay like our “Big Pain” panel.
Table 1: “Big Pain” Panel LC-MS/MS Method Conditions
| Column | Raptor Biphenyl, 100 x 2.1 mm, 2.7 µm (cat.#9309A12) | ||
| Guard Column | Raptor Biphenyl EXP guard column cartridge, 5 x 2.1 mm, 2.7 µm (cat.# 9309A0252) | ||
| Mobile Phase A | Water, 0.1% formic acid, 2 mM ammonium formate | ||
| Mobile Phase B | Methanol, 0.1% formic acid, 2 mM ammonium formate | ||
| Diluent | Water | ||
| Column Temperature | 30°C | ||
| Injection Volume | 5 µL | ||
| Flow Rate | 0.6 mL/min | ||
| Gradient | Time (min) | %A | %B |
| 0.00 | 95 | 5 | |
| 9.00 | 0 | 100 | |
| 10.00 | 0 | 100 | |
| 10.01 | 95 | 5 | |
| 12.00 | 95 | 5 | |
Figure 1: 100 ng/mL of Cychlorphine and Spirochlorphine Using “Big Pain” Method Conditions
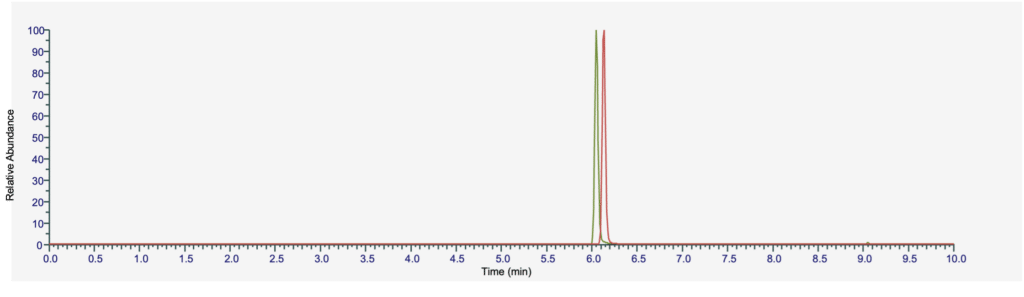
Table 2: MRM Transitions for Cychlorphine and Spirochlorphine
| Analyte | TR (min) | Precursor Ion | Product Ion | Product Ion | Ion Mode |
| Cychlorphine | 6.05 | 409.1 | 271.0 | 138.9 | ESI + |
| Spirochlorphine | 6.13 | 370.1 | 232.1 | 126.9 | ESI + |
Analyzed in ESI positive mode, both analytes will form a precursor ion of [M+H]+. While precursor and product ions should always be optimized for each LC-MS/MS system, the MRM transitions shown above are commonly observed for these compounds and may be used as a starting point for your own method development. Both compounds were found to elute just after six minutes using the described method conditions. Fentanyl (6.02 min) is expected to elute near these compounds as it bears some structural similarities to the orphine analogs. For a complete list of compounds and retention times included in this method, please see Multiclass Drug Panel on Raptor Biphenyl by LC-MS/MS.
References
- UNODC Laboratory and Scientific Service Portals, China: Announcement of class scheduling of “nitazene” analogues, July 2025. www.unodc.org/LSS/Announcement/Details/7e29daf9-1d49-45e6-95e7-8ce932bc94e1
- A.J. Krotulski, D.M .Papsun, B.N. Stang, S.E. Walton, B.K. Logan. Public alert: Increase in fatal overdoses linked to novel synthetic opioid N-propionitrile chlorphine (cychlorphine), Center for Forensic Science Research and Education, United States, 2026. https://www.cfsre.org/nps-discovery/public-alerts/increase-in-fatal-overdoses-linked-to-novel-synthetic-opioid-n-propionitrile-chlorphine-cychlorphine
- K.D. Stahl, W. Van Bever, P. Janssen, E.J. Simon, Receptor affinity and pharmacological potency of a series of narcotic analgesic, anti-diarrheal and neuroleptic drugs, Eur. J. Pharmacol., 46 (3) (1977) 199-205. https://doi.org/10.1016/0014-2999(77)90334-X.
- M.M Vandeputte, A.J Krotulski, D.M Papsun, B. K Logan, C.P. Stove, The rise and fall of isotonitazene and brorphine: Two recent stars in the synthetic opioid firmament, J. Anal. Toxicol., 46 (2) (2022) 115–121. https://doi.org/10.1093/jat/bkab082


