Featured Solutions
Unleash Your Performance with RMX GC Columns→
Get enhanced detection, less downtime, and more discovery with our new, next-generation GC columns.




BrightSpec and Restek have partnered to launch SpectrAline™ Cartridges, bringing together BrightSpec’s deep Molecular Rotational Resonance (MRR) expertise and Restek’s world-class manufacturing excellence. Optimized for MRR, SpectrAline™ Cartridges deliver high sensitivity, efficient sample loading, and access to matrices that challenge traditional workflows—expanding analytical capabilities and bringing new structural clarity to detection.
Presentations
Press Conference
Next-Generation RMX GC Columns
Tuesday, March 10, 2:00-3:00 p.m. | Room 216B
Learn More about RMX GC Columns
Learning Lab
Unleash Your Performance with Next-Generation RMX GC Columns
Wednesday, March 11, 10:30-11:00 a.m. | Show Floor
Speakers: Ramkumar Dhandapani
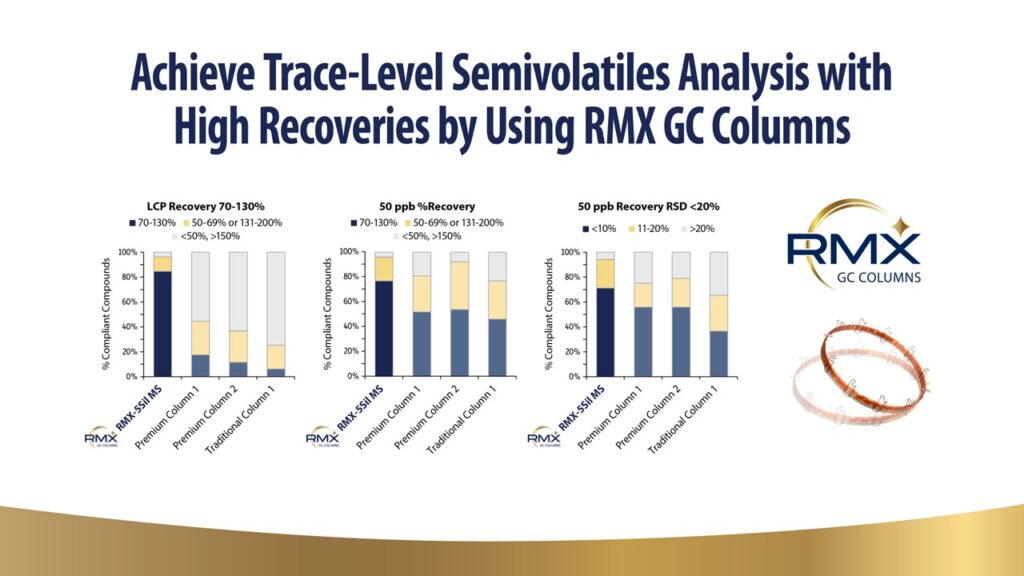
Gas chromatography is a proven technique for the analysis of semivolatiles and compounds. Recent changes in regulations, including a chlorinated solvent ban, method consolidation for pesticides methods, and reduced volume extraction, demanding matrices have lead to a need for a GC solution that can meet and exceed the detection requirements. This presentation will focus on how the latest technology and unique formats in GC can address these challenges.
Organized Sessions
Cannabis Pesticide Testing: Analytical Challenges, Regulatory Gaps, and Opportunities for Improving Consumer Safety
Monday, March 9, 8:30-10:40 a.m. | Room 225B
Speakers: Dan DeLurio
Since the establishment of state-specific medical and recreational cannabis programs, contaminant testing of cannabis and its derivatives has become a critical regulatory requirement intended to protect patients and consumers. Among these regulated contaminants, pesticide residues may pose significant and often under-researched risks to cannabis users who regularly ingest or inhale a wide variety of products available on the market. Currently, pesticide testing regulations vary widely from state to state, with discrepancies in the unique lists of pesticides tested, total number of targets analyzed, and their respective regulatory action limits. This inconsistency has raised concerns among industry experts about the adequacy of pesticide residue testing scopes. In addition to state-to-state variation, there is also growing concern that some states’ pesticide regulations may fail to address the full range of risks associated with cannabis products on the market. Without a harmonized, evolving, and comprehensive testing framework, many consumers may unknowingly be exposed to harmful pesticide residues. This session will explore these challenges within the current testing and regulatory landscape by presenting real-world pesticide data that characterizes ongoing risks to public health. Attendees will gain insight into emerging analytical challenges related to expanding pesticide testing in complex cannabis matrices as well as methods and techniques to overcome these laboratory hurdles. This session calls on laboratories, regulators, and key industry stakeholders to collaborate in advancing pesticide testing to ensure safer, more consistent, higher-quality products for consumers.
Advances in Gas Chromatography to Address Current Chromatographic Challenges
Monday, March 9, 2:30-4:40 p.m. | Room 301B
Speakers: Ramkumar Dhandapani
Method consolidation, compound list increase, ban of chlorinated solvents, and high-sensitive mass spectrometry evolution have all challenged analytical scientists to look at creative solutions. This session will explore innovations in gas chromatography column design and software to tackle these obstacles, including peak shape, sensitivity issues at lower level, matrix resilience, and baseline stability to name a few. We will cover this across four specific topics:
- Role of Surface Deactivation for the Analysis of Low-Level Acidic, Basic, and Neutral Compounds for EPA 8270E
- Cutting Downtime, Not Columns: Performance of a Next-Generation Inert Capillary Column for Multiresidue GC-MSMS Analysis
- Improved Baseline Stability and Sensitivity in GC–MS Using a Novel Column Dimensioned Interface Section
- Virtual Chromatographic Modeling Software for Optimizing Separation of Isobaric Compounds
In summary, this session will highlight how these innovative GC products, product design, methodologies, and software address key gas chromatography challenges, advancing the field and enhancing analytical performance.
Short Course
Method Translation and Modelling in Gas Chromatography (Laptop Required)
Monday, March 9, 1:00-5:00 p.m. | Session SC-7365 | Short Course Office, Room 211
Speakers: Jaap De Zeeuw
In gas chromatography, there is often a wish to maximize uptime of instrumentation. This can be done using higher flows, different column dimensions, a different detector, or a different carrier gas. If you want to get the same peak elution order (same chromatogram), you must make sure that the elution temperatures of components are the same in the new method. This is only possible using a different oven temperature program.
Additionally, it is now also possible to model separations for your analytes that allow you to change all of the parameters using your laptop. There are free, powerful programs available on the web that we will explore. We also will look at GC modeling that you can use for best separation of your components of interest. Using free internet software, you can see the immediate impact of changing the carrier gas, the linear velocity, the stationary phase, the column dimensions, and changing the oven program as well as switching from FID to MS.
For this course, you will have to bring a laptop as you will have to perform the exercises yourself to master these tools.
Oral Presentations
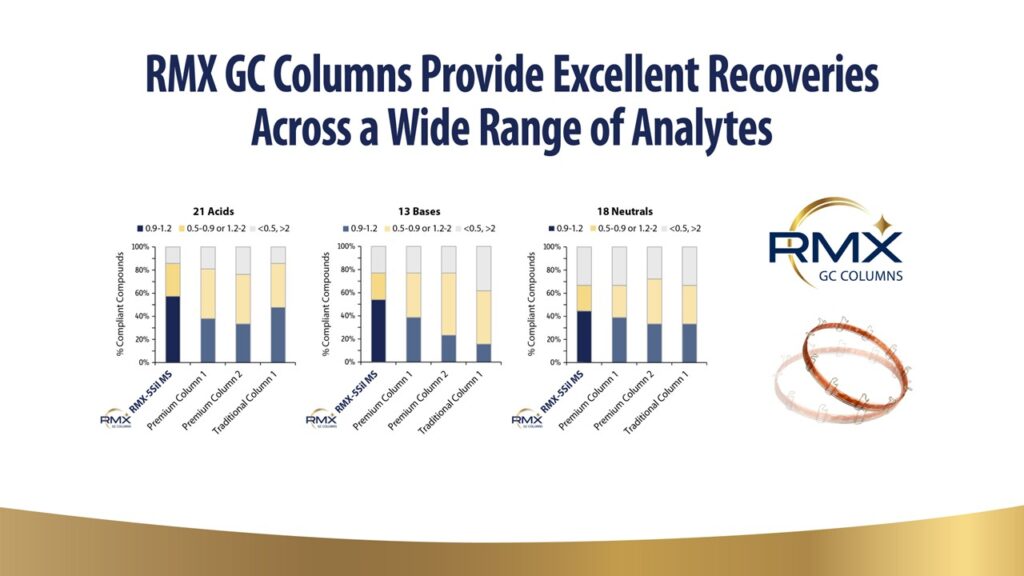
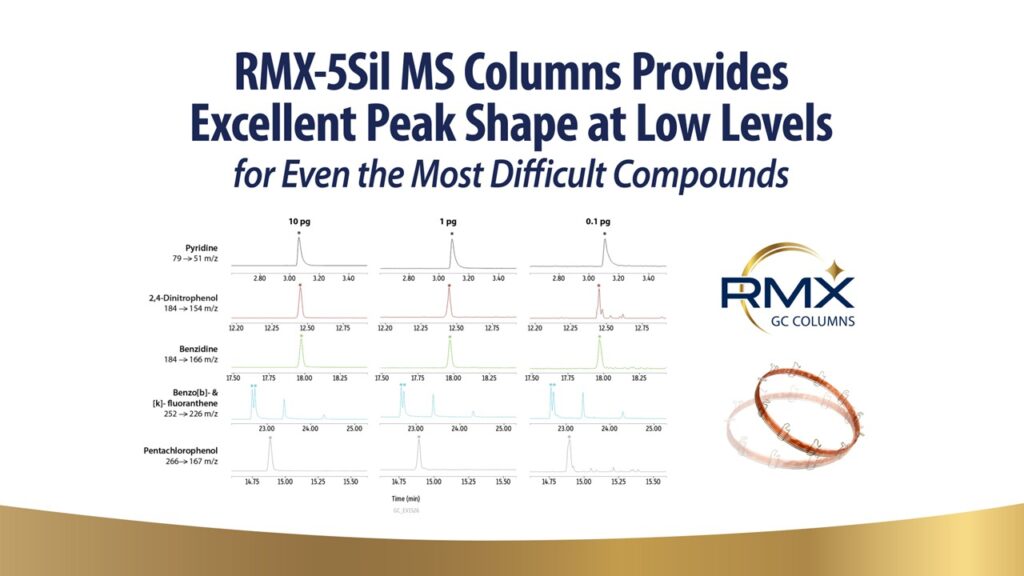
The Role of Surface Deactivation for the Analysis of Low-Level Acidic, Basic, and Neutral Compounds for EPA 8270E
Sunday, March 8, 8:50-9:10 a.m. | Room 305
& Monday, March 9, 3:10-3:30 p.m. | Room 301B
Speakers: Jessi Collier
Environmental laboratories are working towards implementing low-level detection methods using highly sensitive gas chromatography instrumentation, such as gas chromatography tandem triple quadropole mass spectrometry (GC-MS/MS). Reactive surfaces in the flow path of the GC-MS/MS contribute to problems in analyte peak shapes, complicating instrument calibration and consistency. Reagents used in GC column production vary between manufacturers, imparting different types of activity from that surface that becomes more apparent at low levels. Residual silanols at the fused silica surface impart imperfections to the surface of the column, affecting the performance of acids, bases, and neutrals in different ways depending on the chemistry used for deactivation. More information is needed to understand the impact of surface deactivation on low-level analyses where residual activity is exacerbated and more likely to affect productivity. In this study, we evaluated activity of acidic, basic, and neutral compounds associated with the Environmental Protection Agency’s method 8270E, comparing performance at low levels (0.1-10 pg on column). Multiple 5ms columns were evaluated, and the results show how column selection affects analytical performance of acidic, basic, and neutral analytes at low levels.
Improved Baseline Stability and Sensitivity in GC–MS Using a Novel Column with a Unique Interface
Sunday, March 8, 4:40-5:00 p.m. | Room 305
& Monday, March 9, 2:50-3:10 p.m. | Room 301B
Speakers: Ramkumar Dhandapani and Dr. Javier Arrebola (University of Almeria)
A novel gas chromatography–mass spectrometry (GC–MS) column design has been developed to enhance analytical performance by addressing persistent challenges in baseline stability and sensitivity. This innovation integrates a unique interface configuration that minimizes stationary phase degradation and thermal stress effects, particularly under elevated transfer line temperatures. Comparative evaluations using a diverse panel of pesticide analytes demonstrated significantly faster baseline stabilization and reduced background noise relative to conventional columns.
The new column architecture consistently delivered improved signal-to-noise ratios across full-scan and SIM modes with pronounced benefits observed for high-boiling-point and late-eluting compounds. These enhancements suggest a substantial reduction in analyte loss and column bleed, enabling more reliable detection of thermally labile substances.
Improving Detection for Metal-Sensitive Pesticides and Mycotoxins in Cannabis Flower
Monday, March 9, 8:30-9:00 a.m. | Room 225B
Speakers: Melinda Urich
Similar to other crops, cannabis flower can contain contaminants that are harmful to humans, two of the most common being pesticides and mycotoxins. Low limits of detection are necessary for this type of contaminant testing. However, achieving limits of parts per billion can be challenging due to the complexities of cannabis as well as certain analyte properties. These analyte properties include poor ionization and chelation to metal surfaces. Recently, the commercial availability of inert columns has increased in order to address this issue with metal-sensitive compounds seen in traditional stainless steel column hardware. In this work, a selection of pesticides and mycotoxins were chosen for evaluation with attention to both traditionally good and poor-performing analytes. Two separate LC methods, one for pesticides and one for mycotoxins, were developed and used to compare traditional stainless-steel to inert LC hardware for peak shape and sensitivity. Using >15% (w/w) THC content cannabis, samples were prepared using a simple dilute-and-shoot method. In-matrix calibration curves were prepared by post-spiking cannabis extracts with target analytes at various concentrations. Accuracy and precision were assessed. Improvement was notable in both peak shape and peak height when comparing out-of-the-box performance. Results show inert hardware provides a robust analytical solution to improve sensitivity for pesticides and mycotoxins.
Ensuring Quality Data for Organochlorine Pesticide Analysis by Using an Optimized Dual-Bed SPE Cartridge for Sample Extract Cleanup
Monday, March 9, 8:30-11:00 a.m. | Room 305
Speakers: Diego Lopez
One of the most common challenges in analyzing environmental samples for organochlorine pesticides is instrument downtime and shortened calibration periods, both caused by the deleterious effects of coextracted matrix components introduced during sample injection. In addition, chromatographic interferences further complicate analyte identification and quantification, adding to the difficulties faced by environmental analysts.
Ensuring high-quality data depends on maintaining instruments in optimal condition so accurate results can be collected without interference. To achieve this, effective extract cleanup procedures are essential. While traditional sorbents, such as Florisil, silica gel, and alumina, are widely used, these normal-phase options often fail to adequately remove less polar and high molecular weight compounds that compromise instrument performance and sample-path inertness.
In this study, we developed a dual-bed Solid Phase Extraction (SPE) cartridge containing Florisil and Graphitized Carbon Black (GCB) designed to be fully compatible with standard Florisil cleanup workflows. Using this approach, recoveries of all 23 organochlorine pesticides ranged from 89-105% while trichlorophenol was held below 1% in compliance with EPA Method 3620C. The relative standard deviation (RSD) was less than 10% for all analytes.
This newly formulated dual-bed cartridge demonstrates clear advantages when processing highly pigmented and inlet-degrading samples. It enhances the cleanup performance of Florisil alone while minimizing the breakdown of labile analytes, such as endrin and DDT. For analysts, this translates into greater instrument reliability and consistently higher quality data.
Exploring Evolution in Gas Chromatography That Addresses Regulation Needs
Monday, March 9, 2:30-2:50 p.m. | Room 301B
Speakers: Ramkumar Dhandapani
Gas Chromatography is a proven technique for the analysis of semivolatiles and volatile compounds. Recent changes in regulations, including a chlorinated solvent ban, method consolidation, and reduced volume extraction for semivolatiles, has led to a need for a GC solution that can meet and exceed the detection requirements. This presentation will address the challenges in semivolatiles analysis, like EPA 8270 triple and single quad methods, by utilizing the latest innovation in gas chromatography. Unique benefits of novel formats will be explored.
Virtual Chromatographic Modeling Software for Optimizing Separation of Isobaric Compounds
Monday, March 9, 2:30-4:40 p.m. | Room 301B
& Tuesday, March 10, 9:40-10:00 a.m. | Room 305
Speakers: Victoria Zeger
Chromatographic separation modeling tools are becoming increasingly popular for method setup and refinement in high-throughput laboratories looking to minimize instrument downtime. These tools traditionally offer column phase recommendations and customizable method conditions based on the separation of a list of analytes. When using gas chromatography–mass spectrometry (GC-MS), coelutions may be resolved through ion-selective quantitation methods. Shortening method run times often comes at the cost of allowing more coelutions that can successfully be resolved through ion-selective quantitation. However, some methods target separation of isobaric compounds, which cannot be adequately resolved by MS alone and would therefore need to be resolved through column phase chemistry. More user intervention is required to balance phase selection and method parameters for GC-MS method development, delaying the optimization process. As GC-MS techniques continue to be adopted by laboratories, there is a need for more tools that consider mass spectra of analytes when optimizing separations. The virtual chromatographic modeling tool used in this study is fortified with mass spectrum data for compounds included in libraries. When operating in “MS mode,” the software identifies isobaric compounds and prioritizes their separation when recommending column phases and method conditions. The tool is easily toggled to “FID mode” for users interested in resolving all compounds, regardless of mass spectra, allowing for flexibility in instrumentation. Results help labs save time and resources in method development and refinement by considering the instrumentation involved in the model.
Cutting Downtime, Not Columns: Performance of a Next-Generation Inert Capillary Column for Multiresidue GC-MS/MS Analysis
Monday, March 9, 2:30-4:40 p.m. | Room 301B
Speakers: Ally Fairman (Now Foods)
The analysis of pesticide residues in complex botanical matrices is often complicated by high levels of coextracted starches, oils, water, and other naturally occurring compounds that persist even after extensive sample cleanup. Achieving reliable detection and quantitation of pesticides at trace levels demands a chromatographic column with exceptional inertness, resolution, and long-term stability. We present the evaluation of a novel GC capillary column, engineered for outstanding inertness and robustness in GC-MS/MS multiresidue pesticide analysis. Compared to conventional columns, this new design delivers markedly improved peak shape, signal-to-noise, and resolution across a wide variety of challenging botanical matrices, including ashwagandha root, lavender oil, and ginseng powder. A standout feature is its integrated fused silica retention gap at the inlet and fused silica transfer line at the outlet. This innovative construction ensures that retention times remain consistent, even after trimming, allowing for fast and reliable maintenance with no impact on chromatographic performance.
Poster Presentations
Optimizing Injection Port Conditions for the Analysis of Semivolatile Organics
Tuesday, March 10, 1:00-3:00 p.m. | Expo Floor
Speakers: Ramkumar Dhandapani and Victoria Zeger
Split and splitless injections introduce the sample into a heated injection port as a liquid where it is rapidly and completely vaporized along with the analytes in the sample. The vaporized sample condenses at the head of the column along with the solvent. In split injection mode, only a small amount of the vaporized sample is transferred onto the head of the column; the remainder of the sample is removed via the split vent line. The use of split injection minimizes analyte breakdown since compounds spend less time in the hot injection port; however, loss in sensitivity is observed since much of the sample exits the split vent line. In splitless injection, the split valve is closed long enough to allow most of the vaporized sample and solvent in the injection port liner to be transferred to the analytical column, which can take anywhere between 30 and 90 seconds. Since the split vent valve is closed, the sample transfer moves at the speed of the column flow, typically in the 1-2 ml/min range. Solvent focusing in splitless mode requires an oven temperature 20 °C below the boiling point of the sample solvent, allowing the solvent and the analyte to condense and wet the stationary phase. One exception is analyte focusing, which can be performed if the difference between the analyte boiling point and solvent boiling point is 150 °C. There are many analyses that use solvents and compounds where solvent focusing and analyte focusing are not possible. This work will demonstrate optimized conditions for a broad range of semivolatile compounds using methylene chloride as a solvent.
Importance of GC Column Deactivation Technology for the Analysis of Challenging Analytes
Tuesday, March 10, 1:00-3:00 p.m. | PS-E120 | Expo Floor
Speakers: Ramkumar Dhandapani and Jessi Collier
A recent advancement in gas chromatography (GC) column deactivation balances chromatographic performance of acidic, basic, and neutral analytes at trace-level concentrations. Deactivation is a critical step in GC column manufacturing that modifies the fused silica capillary surface activity to promote coating and adhesion. Residual surface silanols, which result from incomplete deactivation or surface contamination, adsorb acidic and basic compounds, resulting in peak area loss or tailing. Deactivation procedures vary between products and manufacturers, with individual benefits and drawbacks that become exacerbated during sensitive applications. There is increasing demand to improve identification and quantification of reactive analytes with complex or consolidated methodologies. New GC deactivation technology improves polymer adhesion and minimizes residual surface activity for a long-lasting, broadly inert surface. Columns built with the new GC deactivation technology were challenged with a wide variety of applications and against traditional 5-type columns. Overall, the new deactivation presents an advancement over traditional GC columns, providing unparalleled inertness to a broader range of acids, bases, and neutrals. These improvements offer opportunity for method consolidation and trace-level analysis that would not be successful with traditional technology.
BrightSpec Presentations
Oral Presentations
Impurity Identification without Reference Standards Using Molecular Rotational Resonance Spectroscopy with BrightSpec CTO Justin Neill
Sunday, March 8, 9:40-10:00 a.m. | Room 304A
The unambiguous structural identification of impurities is a persistent challenge in analytical chemistry. As an example, according to the FDA’s guidance on drug substance impurities (Q3A), any impurity at a level of 0.1% or higher must be structurally identified, including distinguishing among any possible isomeric variants. This process is time-consuming, requiring authentic reference libraries, which may not be readily available. Additionally, multiple techniques are often applied in tandem but can still leave ambiguity in the case of novel impurities or when multiple closely related structures are considered.
We present the development of molecular rotational resonance (MRR) spectroscopy as a standards-free tool that can aid in this often-laborious process. Recent advances in both the sensitivity of MRR measurement, as well as new software workflows, have enabled its application to routine impurity identification. MRR directly characterizes molecules by determining their three-dimensional moments of inertia, giving precise structural information even for compounds that have not yet been experimentally characterized. These parameters can be directly compared to quantum chemistry predictions to allow for confident and rapid identification without requiring reference standards. In addition, MRR’s unique sensitivity to small changes in structure, in particular, stereochemical and regiochemical variations, supports it as an effective complement to other structural identification techniques to address this important need. We will demonstrate this approach on the identification of isomeric impurities in pharmaceutical reaction characterization as well as in other industries.
High-Throughput Impurity Analysis Using Automated Preconcentration and Molecular Resonance Rotational (MRR) Spectroscopy
Monday, March 9, 9:10-9:30 a.m. | Room 304A
Speakers: BrightSpec Senior Application Scientist Alexander Mikhonin
Molecular Resonance Rotational (MRR) Spectroscopy is a novel analytical technique that allows for highly specific detection of molecular species in gas phase, including structural and optical isomers. MRR is highly quantitative and largely matrix-independent, allowing analysis from matrices that are unfriendly to common analytical techniques, like gas chromatography. With the goal of extending to trace detection applications, BrightSpec has successfully coupled MRR spectrometers with fully automated continuous headspace and sample preconcentration techniques, further extending MRR’s analytical range and providing a powerful tool for impurity analysis.
The utility of this system is demonstrated through analysis of volatile analytes that are common impurities in pharmaceutical processes (such as nitrosamines, formaldehyde, ethylene oxide, ethylene and diethylene glycol, and residual solvents). MRR’s capability is demonstrated using both standards and real substances, with an emphasis on selective quantitation of analytes that are challenging to resolve or detect in gas chromatography.
Integration of automated preconcentration and continuous headspace sampling with MRR allows high throughput MRR analysis of these analytes with part-per-billion (ppb) to low part-per-million (ppm) detection limits with minimal sample preparation. Instrument dynamic range is high; signal response is linear over at least three orders of magnitude. The addition of simple offline sample preparation or preconcentration can further improve detection limits.
Poster Presentations
High-Throughput, Fully Automated Analysis of USP<467> Residual Solvents Using Headspace-MRR
Tuesday, March 10, 1:00-3:00 p.m. | PS-P114 | Expo Floor
United States Pharmacopeia (USP) General Chapter <467> “Residual Solvents” recommends static headspace GC (SH-GC) procedures for residual solvents analysis in pharmaceutical products and dietary supplements. However, the standard USP<467> SH-GC procedures are not applicable to all residual solvents. Analytical gaps of SH-GC include low-volatile solvents and water-soluble acids from Classes 2 and 3. A recently published USP Stimuli article demonstrates the ability of Molecular Rotational Resonance (MRR) spectroscopy to bridge the analytical gaps of the conventional static headspace GC for analysis of several GC-unfriendly residual solvents.
This poster extends the scope of the mentioned USP Stimuli article and demonstrates fully automated, high-throughput headspace-MRR analysis of residual solvents in real pharmaceutical substances. The presented method can be readily extended to the analysis of most USP<467> residual solvents as well as the other GC-unfriendly volatile impurities, including, but not limited to, glycols, formaldehyde, ethylene oxide, acetaldehydes, and volatile amines. Further, as MRR is a spectroscopic technique, it can be readily applied to analysis of diverse volatile impurities, including residual solvents, not only at laboratory settings but also at the process floor.
Development of a Spectral Library for Nitrosamine ID and Quantification in Pharmaceuticals and Consumer Products
Tuesday, March 10, 1:00-3:00 p.m. | PS-P110 | Expo Floor
Nitrosamine impurities are a significant concern in pharmaceutical products due to their potential health risks. Commonly used analytical methods (GC/LC, GC/LC-MS, GC/LC-MS/MS) rely on chromatographic separation and advanced sample preparation techniques, which are time-consuming and often have limited ability to distinguish isomeric species. Molecular Rotational Resonance (MRR) spectroscopy is a highly selective spectroscopic technique that provides a fast, reliable detection and quantitation of nitrosamine impurities that is orthogonal to existing techniques. This structure-specific approach allows for confident discrimination between isomers, and BrightSpec’s Automated Spectral Acquisition (ASA) workflow further streamlines the process for development of a spectral library of key nitrosamines of concern.
After identification, targeted quantitation is performed using a single-frequency targeted measurement with integrated preconcentration to improve detection and quantitation limits. BrightSpec’s MRR spectrometers are coupled with a continuous headspace autosampler, allowing direct analysis from matrices unfriendly to GC and LC without complex sample preparation.
The high selectivity of MRR provides unambiguous identification and quantitation of nitrosamine impurities in pharmaceutical products. Coupling of MRR spectrometer to continuous headspace autosampler provides ability to do so from injection unfriendly matrices, like tablets, polymers, etc. Fully automated software reduces user input, making MRR a uniquely practical and powerful technique for pharmaceutical impurity analysis.
Routine Characterization of Enantiomerically Selective Reactions by Molecular Rotational Resonance Spectroscopy
Tuesday, March 10, 1:00-3:00 p.m. | PS-P120 | Expo Floor
The enantiomeric composition of chiral pharmaceuticals is a vital parameter due to the influence of stereochemistry on the efficacy and safety of the product. Despite this, the routine, automated analysis of chiral purity faces significant challenges due to the complexity of method development, usability, and throughput. These challenges create bottlenecks in both R&D and routine quality control, limiting the speed of chiral development.
Molecular Rotational Resonance (MRR) is a powerful spectroscopic technique that addresses long-standing challenges in the analysis of chiral compounds. Enantiomers are resolved through the introduction of a chiral tag, which converts them into diastereomeric complexes that exhibit distinct rotational transitions. Because MRR is highly sensitive to subtle structural differences, the fraction of each enantiomer in a mixture can be quantified without the need for enantiomerically pure reference standards and in the presence of other analytes, eliminating issues with spectral overlap. Importantly, this methodology has proven effective for compound classes that are often difficult to separate by traditional chiral chromatography. Sample introduction and data analysis are straightforward and increasingly automated.
Here, we demonstrate the use of MRR for chiral characterization of 1-phenylethanol, a model system widely studied in asymmetric synthesis. MRR enables unambiguous differentiation of its enantiomers and accurate quantification of enantiomeric excess. Recent advances in automation, software, and spectral libraries have further streamlined method development for new chiral molecules, transforming MRR into a versatile tool for both discovery and process optimization. We demonstrate the advantages of this approach by comparing its analytical performance, simplicity, and throughput to conventional methods.