Solutions for Forensics Analysis
Unleash Your Performance with RMX GC Columns→
Get enhanced detection, less downtime, and more discovery with our new, next-generation GC columns.
Maximize Confidence in Seized Drug Identification by GC-MS→
See excellent chromatographic performance of RMX columns in this GC-MS analysis of 34 drugs of abuse.
Simplify Your GC Method Development with Our Free Pro EZGC Software→
Method Development Guide for Novel Psychoactive Substances→
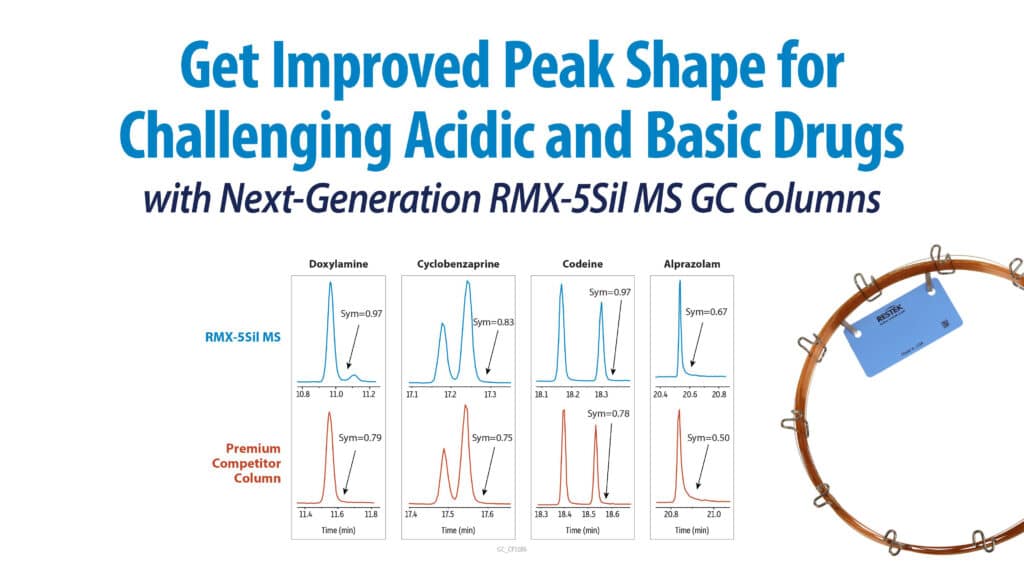
Reduce Downtime and Cost of Materials with Rugged Rxi-5Sil MS GC Columns→
Restek’s Biphenyl LC Column: Two Decades of Pioneering Innovation→
Strategies For Managing High Concentrations of Gabapentin in Urine by LC-MS/MS→
Screening Methods for Drugs of Abuse Compounds Using GC→
A Comparison of Sample Preparation Techniques for Drugs of Abuse in Oral Fluid→
Maximize Performance with Inert LC Columns and Guards→
Fast, Definitive Data for Blood Alcohol Testing on Rtx-BAC Plus 1 and Rtx-BAC Plus 2 Columns→
Presentations
Showcase
Unleash Your Performance with the Next Generation GC Column Technology
Wednesday, February 11, 2:00-2:20 p.m. | Exhibit Floor
Speakers: Amanda Rigdon
Oral Presentations
Strategies for Mitigating the Effects of High Gabapentin Concentrations in Urine Samples
Friday, February 13, 9:15-9:30 a.m. | NOCC 356
Speakers: Haley Berkland, MS
Gabapentin is an anti-convulsant drug that is prescribed for the treatment of neuropathic pain and seizures, as well as for many off label uses. Due to the pharmacokinetics of gabapentin, urine specimens often contain extremely high concentrations of the compound. When analyzed by LC-MS/MS, high concentrations of gabapentin can have significant analytical implications, particularly for the compound amphetamine. Interference between gabapentin and amphetamine has been well documented, and can result in signal suppression, poor peak shape, and shifting retention times. Other analytical challenges include saturation of the mass spectrometry detector and column overload.
In this work, we explored several strategies to mitigate the effects of high gabapentin concentrations in urine samples. A method used in the analysis of 60 drugs of abuse in urine was used to test samples containing varying concentrations of gabapentin and amphetamine. After data was collected, the signal of amphetamine was compared in each sample to determine if interference from gabapentin was occurring. The method utilized a Raptor Biphenyl 50 x 2.1 mm, 2.7 µm column and a mobile phase of water and methanol, both acidified with 0.1% formic acid. The flow rate was 0.6 mL/min, the column temperature was 45 °C, and the injection volume was 5 µL. Gradient elution was employed. In samples containing high amounts of gabapentin, amphetamine showed decreased sensitivity due to signal suppression and shifted retention time. Different strategies were investigated to see if the interference between gabapentin and amphetamine could be resolved. These strategies included using alternate column lengths and diameters, decreasing injection volume, and testing different mobile phase additives.
It was determined that the best approach for reducing interference was to fully chromatographically resolve gabapentin and amphetamine and ensuring that gabapentin was the first compound to elute. This was accomplished by switching the additive in mobile phase A from formic acid to ammonium formate, which impacted the elution order of early eluting compounds. Resolution was further improved by switching a 100 x 2.1 mm column. Detector saturation and column overload were mitigated by reducing the injection volume from 5 µL to 2 µL. Significant carryover was observed due to the high analyte concentrations and was eliminated by adding a small amount of 2-propanol to mobile phase B. Performance of the other analytes in the method was not negatively affected by the change in method parameters. The redeveloped method can be used to effectively analyze 60 drugs of abuse in urine without interference between gabapentin and amphetamine.
Analyzing the Impact of Centrifugation and Sample Preparation on Drugs of Abuse in Oral Fluids
Friday, February 13, 9:45-10:00 a.m. | NOCC 356
Speakers: Samantha Herbick, MS
Oral fluid is becoming an increasingly popular matrix due to its ease of collection. While oral fluid is a relatively simple matrix to collect, the collection kits can cause issues downstream. Kits often consist of an applicator with a sponge attached, a tube filled with 3 mL of buffer solution, and a cap. The sponge is used to collect oral fluid which is then placed in the buffer solution to ensure analyte stability and inhibit bacterial growth. The total volume of solution for testing should be 4 mL (1 mL of oral fluid and 3 mL of buffer), however, it is often very difficult to recover the full volume of solution, which can affect volume and analyte recovery. There are different techniques to manipulate the sponge and improve sample recovery, such as manual compression and centrifugation. Additionally, the buffers used with the kits contain surfactants that can lead to issues downstream such as matrix interferences and shortened column lifetimes, so it is important to remove these buffers during sample preparation. In this work, two sample removal techniques will be compared by examining volume and analyte recovery, as well as highlighting the benefits of sample preparation when analyzing oral fluid samples.
An LC-MS/MS method was developed using a biphenyl analytical column and mobile phases consisting of water, and methanol both containing 0.1% formic acid. A total of 31 common drugs of abuse were separated under gradient conditions, with a cycle time of 7 minutes. The samples were prepared in synthetic oral fluid and combined with Quantisal buffer and kits. Two recovery techniques were compared and tested in triplicate: manual compression and centrifugation. Samples were prepared using a salt-assisted liquid-liquid extraction (SALLE).
The two techniques were compared by examining volume and analyte recovery. Volume recovery was tested by pouring the solution into a graduated cylinder and recording the total volume recovered using each technique. When using centrifugation, on average, an additional 200 μL were collected compared to the manual compression. Analyte recovery was compared by spiking a known concentration, 50 ng/mL, into each of the samples. These samples were evaluated using a calibration curve prepared only using 1 mL of fortified synthetic oral fluid and 3 mL of buffer. The recovery and peak area of all analytes improved when using the centrifuge. Accuracy and precision were also compared by analyzing if the results fell within +/- 15% of the target value as well as comparing %RSD values. The use of extraction aids was also investigated and compared to these benchmarks.
Through examining volume recovery and analyte recovery when using the two techniques, it was clear that the centrifugation technique showed the best results for total volume recovered as well as increased analyte recovery when analyzed quantitatively. This work clearly demonstrates that centrifugation paired with efficient sample clean up not only improves analyte sensitivity but also lengthens the lifetime of the column when analyzing drugs of abuse in oral fluid.
Poster Presentations
Semisynthetic Cannabinoids: The Newest Challenge in Analyzing THC Isomers
Wednesday, February 11, 7:30-9:00 a.m. | Hilton Fulton
Speakers: Haley Berkland, MS
Download
Over the past several years, Δ8-THC has made headlines as a popular “legal” alternative to Δ9-THC. The rise in popularity of Δ8-THC led to a new analytical challenge for toxicology laboratories—differentiating between the Δ8 and Δ9 isomer forms. For labs performing this testing by LC-MS/MS, complete separation of these isomers and their metabolites is necessary for accurate quantitation.
Recently, a new group of compounds referred to as semi-synthetic cannabinoids have emerged on the market. Drug monitoring groups in the U.S. have suggested that these compounds be added to toxicology testing scopes due to increased use and detection.
Many toxicology laboratories have already implemented LC-MS/MS methods to simultaneously analyze for Δ8-THC and Δ9-THC in casework. In this work, six emerging semi-synthetic cannabinoids were added to an existing LC-MS/MS used for the analysis of Δ8-THC, Δ9-THC, and their hydroxy and carboxy metabolites in whole blood. The six cannabinoids (9(R)-HHC, 9(S)-HHC, Δ10-THC, THC-O-Acetate, THCP, and CBDP) were all identified as scope recommendations in early 2025.
The six semi-synthetic cannabinoids were run on the original method conditions. The method used a Raptor FluoroPhenyl 100 x 3 mm, 2.7 µm column and mobile phases of water and methanol, both acidified with 0.1% formic acid. The column temperature was 40°C, the flow rate was 0.8 mL/min, and gradient elution was used. While resolution was achieved for all compounds, including isomers, using the original method conditions, THCP and THC-O-Acetate eluted at 100% B, where matrix interferences are known to wash off the column. To prevent interference, the method conditions were altered to allow these compounds more time on the column. Additional resolution for the isomers was also achieved by dropping the column temperature from 40 °C to 30 °C. All six semi-synthetic cannabinoids were successfully integrated into a pre-existing LC-MS/MS method for the analysis of THC isomers in whole blood.
A Rapid LC-MS/MS Analysis of EtG/EtS in Urine
Wednesday, February 11, 7:30-9:00 a.m. | Hilton Fulton
Speakers: Jamie York, PhD, Samantha Herbick, MS
Download
Ethyl Glucuronide (EtG) and Ethyl Sulfate (EtS) are established biomarkers for alcohol consumption. The analysis of these compounds poses challenges due to their polar nature, making them challenging to retain by reversed phase chromatography. Additionally, isobaric matrix interferences in urine must be fully resolved from the analytes to obtain accurate data.
In this work, a rapid LC-MS/MS method was developed that provides excellent resolution between the matrix interferences and the analytes of interest. The goal of this study was to establish a robust method capable of retaining both EtG and EtS while resolving urinary matrix interferences without the need for time consuming column pre-conditioning.
Synthetic urine was fortified with EtG and EtS from 50-1,000 ng/mL. Quality control (QC) samples were prepared using six lots of human urine, including samples from kidney disease and liver disease patients. Human urine was diluted 20-fold in water fortified with 0.1% formic acid. The samples were vortexed and centrifuged prior to injection on a Force Biphenyl column (100 x 3 mm, 3 μm). The mobile phases used were 0.1% formic acid in both water and methanol and the chromatographic separation was achieved under gradient conditions.
The developed method successfully resolved matrix interferences from the analytes of interest and no significant matrix interferences were observed across all QC urine lots. Linearity was demonstrated for both analytes with r² ≥ 0.99, and the method exhibited acceptable intra- and inter-day precision and accuracy.
The Force Biphenyl column offered improved robustness over multi-mode columns, which often require extensive pre-conditioning. In contrast, the Force Biphenyl allowed immediate use without compromising performance. It also provides flexibility for analyzing a broad range of compounds, including drugs of abuse and novel psychoactive substances, reducing the need for column changes.
Evaluation of Deactivations on Gas Chromatographic Stationary Phases for the Analysis of Seized Drugs of Abuse
Thursday, February 12, 11:30-1:00 p.m. | Hilton Fulton
Speakers: Amanda Rigdon, Corby Hilliard
Download
Gas Chromatography-Mass Spectrometry (GC-MS) often used for screening seized drugs due to its ease of use and specificity. This technique is particularly advantageous because it can be performed without the need for chemical derivatization, a process that is often time-consuming and complex. However, when drugs of abuse are analyzed in their underivatized form, their active functional groups can interact with the active sites on the surface of the fused silica column. This interaction can result in poor peak shape and reduced recoveries, potentially leading to inaccurate or unreliable results.
Recent advancements in column chemistry, particularly the development of deactivations that minimize the presence of active silanols on the fused silica surface, have significantly improved the ability to analyze active compounds without derivatization. Given the highly reactive nature of the compounds involved, column deactivation plays a crucial role in achieving reliable results.
In this study, we evaluated the impact of column deactivation on a 5% diphenyl-polysiloxane stationary phase (5-type) to identify the optimal deactivation for analyzing a broad range of underivatized drugs which includes compounds that contain acidic, basic, and neutral moieties. As the complexity of drugs of abuse continues to increase, laboratories are challenged to implement more comprehensive lists of compounds in their methods, ensuring they can detect a wider range of substances, including new psychoactive compounds (NPS).